Tanespimycin (17-N-allylamino-17-demethoxygeldanamycin, 17-AAG) is a derivative of the antibiotic geldanamycin that is being studied in the treatment of cancer, specifically in younger patients with certain types of leukemia or solid tumors, especially kidney tumors.
| |
| Names | |
|---|---|
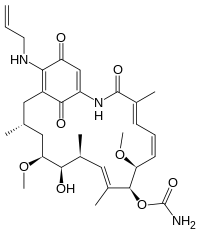
| IUPAC name
[(3S,5S,6R,7S,8E,10R,11S,12E,14E)-21-(allylamino)-6-hydroxy-5,11-dimethoxy-3,7,9,15-tetramethyl-16,20,22-trioxo-17-azabicyclo[16.3.1]docosa-8,12,14,18,21-pentaen-10-yl] carbamate
| |
| Other names
17-N-Allylamino-17-demethoxygeldanamycin
17-AAG | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C31H43N3O8 | |
| Molar mass | 585.698 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
It works by inhibiting Hsp90, which is expressed in those tumors.[1]
It belongs to the family of drugs called antitumor antibiotics.
Clinical trials
editBristol-Myers Squibb conducted Phase 1[2][3] and Phase 2 clinical trials. However, in 2010 the company halted development of tanespimycin, during late-stage clinical trials as a potential treatment for multiple myeloma. While no definitive explanation was given, it has been suggested that Bristol-Myers Squibb halted development over concerns of the financial feasibility of tanespimycin development given the 2014 expiry of the patent on this compound, and the relative expense of manufacture.[4]
References
edit- ^ Dimopoulos MA, Mitsiades CS, Anderson KC, Richardson PG (February 2011). "Tanespimycin as antitumor therapy". Clinical Lymphoma, Myeloma & Leukemia. 11 (1): 17–22. doi:10.3816/CLML.2011.n.002. PMID 21454186.
- ^ Clinical trial number NCT00093821 for "Phase 1 trial: 17-N-Allylamino-17-Demethoxygeldanamycin (17-AAG) in Treating Young Patients With Recurrent or Refractory Leukemia or Solid Tumors" at ClinicalTrials.gov
- ^ Clinical trial number NCT00079404 for "Phase 1 trial: 17-N-Allylamino-17-Demethoxygeldanamycin in Treating Young Patients With Relapsed or Refractory Solid Tumors or Leukemia" at ClinicalTrials.gov
- ^ "Bristol-Myers Squibb Halts Development of Tanespimycin". The Myeloma Beacon. 22 July 2010. Archived from the original on 28 December 2010.