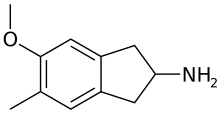
5-Methoxy-6-methyl-2-aminoindane (MMAI) is a drug of the 2-aminoindane group developed in the 1990s by a team led by David E. Nichols at Purdue University.[1] It acts as a less neurotoxic and highly selective serotonin releasing agent (SSRA) and produces entactogenic effects in humans.[1][2][3][4] It has been sold as a designer drug and research chemical online since 2010.
 | |
| Clinical data | |
|---|---|
| Other names | MMAI; MMAi; 5-Methoxy-6-methyl-2-aminoindan |
| Routes of administration | By mouth |
| Drug class | Selective serotonin releasing agent; Entactogen |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C11H15NO |
| Molar mass | 177.247 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
MMAI has been shown to relieve stress-induced depression in rats more robustly than sertraline,[5] and as a result it has been suggested that SSRAs like MMAI and 4-methylthioamphetamine (4-MTA) could be developed as novel antidepressants with a faster onset of therapeutic action and superior effectiveness to current antidepressants such as the selective serotonin reuptake inhibitors (SSRIs).[6]
MMAI alone does not appear to produce serotonergic neurotoxicity with either acute or chronic administration in animals.[7][8] However, subsequent research found that a single high dose of MMAI could produce significant serotonergic neurotoxicity.[7][8] In addition, combination of MMAI with the dopamine releasing agent dextroamphetamine has been found to produce dose-dependent serotonergic neurotoxicity in animals.[7] Hence, MMAI is not a fully non-neurotoxic MDMA analogue.[7][8]
References
edit- ^ a b Marona-Lewicka D, Nichols DE (June 1994). "Behavioral effects of the highly selective serotonin releasing agent 5-methoxy-6-methyl-2-aminoindan". European Journal of Pharmacology. 258 (1–2): 1–13. CiteSeerX 10.1.1.688.1895. doi:10.1016/0014-2999(94)90051-5. PMID 7925587.
- ^ Li Q, Murakami I, Stall S, Levy AD, Brownfield MS, Nichols DE, Van de Kar LD (December 1996). "Neuroendocrine pharmacology of three serotonin releasers: 1-(1,3-benzodioxol-5-yl)-2-(methylamino)butane (MBDB), 5-methoxy-6-methyl-2-aminoindan (MMAi) and p-methylthioamphetamine (MTA)". The Journal of Pharmacology and Experimental Therapeutics. 279 (3): 1261–1267. PMID 8968349.
- ^ Rudnick G, Wall SC (February 1993). "Non-neurotoxic amphetamine derivatives release serotonin through serotonin transporters". Molecular Pharmacology. 43 (2): 271–276. PMID 8429828.
- ^ Luethi D, Kolaczynska KE, Docci L, Krähenbühl S, Hoener MC, Liechti ME (May 2018). "Pharmacological profile of mephedrone analogs and related new psychoactive substances" (PDF). Neuropharmacology. 134 (Pt A): 4–12. doi:10.1016/j.neuropharm.2017.07.026. PMID 28755886. S2CID 28786127.
- ^ Marona-Lewicka D, Nichols DE (December 1997). "The Effect of Selective Serotonin Releasing Agents in the Chronic Mild Stress Model of Depression in Rats". Stress. 2 (2): 91–100. doi:10.3109/10253899709014740. PMID 9787258.
- ^ Scorza C, Silveira R, Nichols DE, Reyes-Parada M (July 1999). "Effects of 5-HT-releasing agents on the extracellullar hippocampal 5-HT of rats. Implications for the development of novel antidepressants with a short onset of action". Neuropharmacology. 38 (7): 1055–1061. doi:10.1016/S0028-3908(99)00023-4. PMID 10428424. S2CID 13714807.
- ^ a b c d Johnson MP, Nichols DE (July 1991). "Combined administration of a non-neurotoxic 3,4-methylenedioxymethamphetamine analogue with amphetamine produces serotonin neurotoxicity in rats". Neuropharmacology. 30 (7): 819–822. doi:10.1016/0028-3908(91)90192-e. PMID 1717873.
- ^ a b c Johnson MP, Conarty PF, Nichols DE (July 1991). "[3H]monoamine releasing and uptake inhibition properties of 3,4-methylenedioxymethamphetamine and p-chloroamphetamine analogues". Eur J Pharmacol. 200 (1): 9–16. doi:10.1016/0014-2999(91)90659-e. PMID 1685125.