Flmodafinil (developmental code names CRL-40,940, NLS-4, JBG01-41), also known as bisfluoromodafinil and lauflumide, is a wakefulness-promoting agent related to modafinil which has been developed for treatment of a variety of different medical conditions.[2][3][4] These include chronic fatigue syndrome, idiopathic hypersomnia, narcolepsy, attention deficit hyperactivity disorder (ADHD), and Alzheimer's disease.[3][4] Aside its development as a potential pharmaceutical drug, flmodafinil is sold online and used non-medically as a nootropic (cognitive enhancer).[5][6][7]
 | |
| Clinical data | |
|---|---|
| Other names | CRL-40,941; CRL-40941; NLS-4; JBG01-41; JBG1-41; JBG1-041; Bisfluoromodafinil; Lauflumide; JBG1-048/JBG1-049; JBG01-048/JBG01-049[1] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
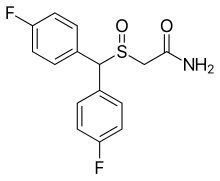
| Formula | C15H13F2NO2S |
| Molar mass | 309.33 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
The drug has been found to act as a selective atypical dopamine reuptake inhibitor.[2][8][1][9] It produces wakefulness-promoting effects in animals.[2][8] Unlike modafinil, flmodafinil does not induce cytochrome P450 enzymes.[2] Chemically, flmodafinil is an enantiopure derivative of modafinil and is also known as bisfluoromodafinil (it is the (R)-bis(4-fluoro) phenyl ring-substituted derivative of modafinil).[2][8]
Flmodafinil was developed by NLS Pharma.[3] As of January 2024, it is in preclinical development for treatment of chronic fatigue syndrome.[3] No recent development has been reported for idiopathic hypersomnia and development has been discontinued for narcolepsy, ADHD, and Alzheimer's disease.[3]
Pharmacology
editPharmacodynamics
editFlmodafinil is a selective dopamine reuptake inhibitor (DRI).[2][8][10] Its affinity (Ki) for the DAT is 4,090 nM.[10] At the serotonin transporter (SERT), its affinity (Ki) was 48,700 nM (12-fold lower than for the DAT), and it had negligible affinity for the sigma σ1 receptor (Ki > 100,000 nM).[10] The drug has been found to block the dopamine transporter (DAT) by 83%, to a greater extent than methylphenidate without unfavorable concomitant adrenergic effects.[8] The drug is an atypical DRI similarly to modafinil.[11][1][9]
The affinities for the DAT of flmodafinil's enantiomers and modafinil have also been studied.[1][10] The affinities (Ki) were 5,480 nM for armodafinil ((R)-modafinil), 2,970 nM for (S)-(+)-flmodafinil (JBG1-048), and 4,830 nM for (R)-(–)-flmodafinil (JBG1-049).[1][10] Their affinities for the SERT and σ1 receptor have also been reported.[10] Similarly to modafinil, (S)-(+)-flmodafinil and (R)-(–)-flmodafinil increase dopamine levels in the nucleus accumbens in animals.[1] They have been found to increase dopamine levels by up to 150 to 200% of baseline at the highest assessed dose.[1] These increases are much smaller than those elicited by amphetamine or cocaine.[1][12]
In a study comparing the wake-promoting effects of flmodafinil and modafinil, flmodafinil was found to maintain wakefulness over a significantly longer timeframe than modafinil.[8] While the administration of neither compound resulted in sleep rebound, flmodafinil perturbed sleep architecture to a lesser degree than modafinil.[8] This difference was characterised by an attenuated EEG power density within slow frequencies (<4 Hz) following flmodafinil treatment, though both compounds increased power density relative to placebo.[8]
In contrast to modafinil, flmodafinil is not an inducer of the cytochrome P450 CYP3A4 or CYP3A5 enzymes.[2]
Chemistry
editFlmodafinil is a racemic mixture of (S)-(+)- and (R)-(–)-enantiomers.[11][1] The (S)-(+) enantiomer has been referred to as JBG1-048 and the (R)-(–) enantiomer has been referred to as JBG1-049.[1]
Analogues of flmodafinil include modafinil, armodafinil ((R)-modafinil), esmodafinil ((S)-modafinil), adrafinil (CRL-40,028; N-hydroxymodafinil), fladrafinil (CRL-40,941; bisfluoroadrafinil), and CE-123, among others.
History
editFlmodafinil was patented in 2013, and preclinical research has been underway since December 2015.[3][4][13][14][15][16] It appears to have first been patented in the 1980s.[17][13]
Research
editThe pharmacokinetics of flmodafinil are being studied.[18]
References
edit- ^ a b c d e f g h i j Keighron JD, Giancola JB, Shaffer RJ, DeMarco EM, Coggiano MA, Slack RD, et al. (August 2019). "Distinct effects of (R)-modafinil and its (R)- and (S)-fluoro-analogs on mesolimbic extracellular dopamine assessed by voltammetry and microdialysis in rats". Eur J Neurosci. 50 (3): 2045–2053. doi:10.1111/ejn.14256. PMC 8294075. PMID 30402972.
- ^ a b c d e f g Konofal E (August 2024). "From past to future: 50 years of pharmacological interventions to treat narcolepsy". Pharmacol Biochem Behav. 241: 173804. doi:10.1016/j.pbb.2024.173804. PMID 38852786.
Among these advancements is lauflumide (NLS-4), a forward step from earlier substances initially envisioned by Lafon Laboratories yet not realized (Dowling et al., 2017). Developed by NLS Pharmaceutics AG, lauflumide represents a cutting-edge development as a selective dopamine reuptake inhibitor. It is an enantiomerically pure R-isomer, with an enantiomeric excess exceeding 95 %, of a bis(p-fluoro) phenyl ring-substituted derivative of modafinil, showcasing the innovative work of inventor Eric Konofal (USPTO Patent 2017, US9637447B2) (Konofal, 2017). Unlike modafinil, which induces hepatic enzyme activity with repeated doses, lauflumide does not act as an inducer of cytochrome P450 (CYP) enzymes, including CYP3A4/5. In mouse models, lauflumide has demonstrated potent wake-promoting effects without the risk of hypersomnia rebound (unpublished data). Moreover, the recovery sleep following lauflumide administration is marked by a reduced amount of NREM sleep and delta wave activity, indicating a decreased need for recovery sleep despite extended periods of wakefulness induced by the drug (Luca et al., 201 ).
- ^ a b c d e f "Lauflumide - NLS Pharmaceutics Ltd". AdisInsight. Springer Nature Switzerland AG. 28 January 2024. Retrieved 17 August 2024.
- ^ a b c "Pipeline". monsolfoundation.ch. 30 September 2022. Retrieved 17 August 2024.
- ^ Sousa A, Dinis-Oliveira RJ (2020). "Pharmacokinetic and pharmacodynamic of the cognitive enhancer modafinil: Relevant clinical and forensic aspects". Subst Abus. 41 (2): 155–173. doi:10.1080/08897077.2019.1700584. PMID 31951804.
Modafinil is a highly researched compound, with many analogues created and studied (Figure 1); the wakefulness promoting agents CRL-40,490 and modafiendz are the fluoro and N-methyl analogs of modafinil and the CRL-40,491 is the fluoro analog of adrafinil.30,31 [...] Although the long-term effects in healthy individuals are unknown, modafinil is easily available online with limited information about the use of and potential harms related to the drug.20,209 Other possibilities available from online shops and other retail outlets include adrafinil, CRL-40,940, CRL40,941 and modafiendz.30 Alternatively to online purchase, students also report to obtain stimulants from a pharmacy with or without prescription, from colleagues, friends or family, or from an herbalist.20
- ^ Schifano F, Catalani V, Sharif S, Napoletano F, Corkery JM, Arillotta D, et al. (April 2022). "Benefits and Harms of 'Smart Drugs' (Nootropics) in Healthy Individuals". Drugs. 82 (6): 633–647. doi:10.1007/s40265-022-01701-7. PMID 35366192.
[Modafinil] is widely available for online purchase [105] and it is of interest that a range of modafinil derivatives are actively being discussed on web fora, including: adrafinil, fladrafinil, flmodafinil, and N-methyl-4,4′-difluoro-modafinil [8]. Finally, the modafinil R-enantiomer armodafinil, which is being used to improve wakefulness in patients with excessive sleepiness [106], is currently the subject of an anecdotal debate relating to its properties as a [cognitive enhancer] [107].
- ^ Dowling G, Kavanagh PV, Talbot B, O'Brien J, Hessman G, McLaughlin G, et al. (March 2017). "Outsmarted by nootropics? An investigation into the thermal degradation of modafinil, modafinic acid, adrafinil, CRL-40,940 and CRL-40,941 in the GC injector: formation of 1,1,2,2-tetraphenylethane and its tetra fluoro analog" (PDF). Drug Test Anal. 9 (3): 518–528. doi:10.1002/dta.2142. PMID 27928893.
- ^ a b c d e f g h Luca G, Bandarabadi M, Konofal E, Lecendreux M, Ferrié L, Figadère B, et al. (2018). "Lauflumide (NLS-4) Is a New Potent Wake-Promoting Compound". Front Neurosci. 12: 519. doi:10.3389/fnins.2018.00519. PMC 6104159. PMID 30158846.
Preliminary findings suggest that NLS-4 is a selective dopamine reuptake inhibitor, blocking (83%) dopamine transporter (DAT), higher than methylphenidate and without deleterious effects on peripheral adrenergic systems involved in hypertension (Study 100014859 CEREP 20/03/14, unpublished data).
- ^ a b Zanettini C, Scaglione A, Keighron JD, Giancola JB, Lin SC, Newman AH, et al. (December 2019). "Pharmacological classification of centrally acting drugs using EEG in freely moving rats: an old tool to identify new atypical dopamine uptake inhibitors". Neuropharmacology. 161: 107446. doi:10.1016/j.neuropharm.2018.11.034. PMC 8369976. PMID 30481526.
The atypical DUI modafinil and its F-analog, JBG1-049, decreased the power of beta, but in contrast to cocaine, none of the other frequency bands, while JHW007 did not significantly alter the EEG spectrum. [...] Comparative analysis of the effects of test drugs on EEG indicates a potential atypical profile of JBG1-049 with similar potency and effectiveness to its parent compound modafinil. [...] In summary, quantitative analysis of EEG spectra revealed different neurosignatures of typical and atypical DUIs and of other central nervous system active drugs. These data suggest that evaluation of the EEG signal can be used to identify new DUIs, such as JBG 1-049, with potential atypical profiles in the early preclinical phase of drug development and can accelerate the discovery of possible treatments for psychostimulant use disorders.
- ^ a b c d e f Giancola JB, Bonifazi A, Cao J, Ku T, Haraczy AJ, Lam J, et al. (December 2020). "Structure-activity relationships for a series of (Bis(4-fluorophenyl)methyl)sulfinylethyl-aminopiperidines and -piperidine amines at the dopamine transporter: Bioisosteric replacement of the piperazine improves metabolic stability". European Journal of Medicinal Chemistry. 208: 112674. doi:10.1016/j.ejmech.2020.112674. PMC 7680422. PMID 32947229.
- ^ a b Tanda G, Hersey M, Hempel B, Xi ZX, Newman AH (February 2021). "Modafinil and its structural analogs as atypical dopamine uptake inhibitors and potential medications for psychostimulant use disorder". Curr Opin Pharmacol. 56: 13–21. doi:10.1016/j.coph.2020.07.007. PMC 8247144. PMID 32927246.
JBG1–048 and JBG1–049, two bis F-MOD enantiomers, administered intravenously, produced dose-dependent increases in DA efflux similar to, but longer lasting than those elicited by (R)-MOD [45] or MOD [42].
- ^ Ramsson ES, Howard CD, Covey DP, Garris PA (December 2011). "High doses of amphetamine augment, rather than disrupt, exocytotic dopamine release in the dorsal and ventral striatum of the anesthetized rat". J Neurochem. 119 (6): 1162–1172. doi:10.1111/j.1471-4159.2011.07407.x. PMC 3213283. PMID 21806614.
- ^ a b CA 1199916, Lafon L, "Benzhydrylsulfinylacetamide derivatives", published 28 January 1986, issued 28 January 1986, assigned to Cephalon France SAS
- ^ Cao J, Prisinzano TE, Okunola OM, Kopajtic T, Shook M, Katz JL, et al. (October 2010). "Structure-Activity Relationships at the Monoamine Transporters for a Novel Series of Modafinil (2-[(diphenylmethyl)sulfinyl]acetamide) Analogues". ACS Medicinal Chemistry Letters. 2 (1): 48–52. doi:10.1021/ml1002025. PMC 3041981. PMID 21344069.
- ^ US 20130295196, Konofal E, "Lauflumide and the enantiomers thereof, method for preparing same and therapeutic uses thereof", published 7 November 2013, issued 2 May 2017, assigned to NLS Pharmaceutics AG and Assistance Publique Hopitaux de Paris APHP
- ^ US 4489095, Lafon L, "Halogenobenzhydrylsulfinylacetohydroxamic acids", issued 18 December 1984, assigned to Cephalon France SAS
- ^ "Acetamide, 2-[[bis(4-fluorophenyl)methyl]sulfinyl]-". PubChem. Retrieved 18 August 2024.
- ^ "Investigations into the metabolism and elimination of flmodafinil and fladrafinil as well as their prevalence in elite sports". World Anti Doping Agency. 23 May 2024. Retrieved 18 August 2024.