Dichlorine heptoxide is the chemical compound with the formula Cl2O7. This chlorine oxide is the anhydride of perchloric acid. It is produced by the careful distillation of perchloric acid in the presence of the dehydrating agent phosphorus pentoxide:[1]

| |
| |
| Names | |
|---|---|
| IUPAC name
Dichlorine heptoxide
| |
| Other names
Chlorine(VII) oxide; Perchloric anhydride; (Perchloryloxy)chlorane trioxide
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| Cl2O7 | |
| Molar mass | 182.901 g/mol |
| Appearance | colorless liquid, colorless gas |
| Density | 1.9 g/cm3 |
| Melting point | −91.57 °C (−132.83 °F; 181.58 K) |
| Boiling point | 82.07 °C (179.73 °F; 355.22 K) |
| hydrolyzes to form perchloric acid | |
| Thermochemistry | |
Std enthalpy of
formation (ΔfH⦵298) |
275.7 kJ/mol |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
oxidizer, contact explosive[1] |
| NFPA 704 (fire diamond) | |
| Related compounds | |
Related compounds
|
Manganese heptoxide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
- 2 HClO4 + P4O10 → Cl2O7 + H2P4O11
The chlorine(VII) oxide can be distilled off from the mixture.
It may also be formed by illumination of mixtures of chlorine and ozone with blue light.[2] It slowly hydrolyzes back to perchloric acid.
Structure
editCl2O7 is an endergonic molecule, meaning it is intrinsically unstable, decomposing to its constituent elements with release of energy:[3]
- 2 Cl2O7 → 2 Cl2 + 7 O2 (ΔH° = –132 kcal/mol)
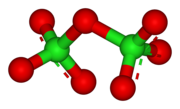
Dichlorine heptoxide is a covalent compound consisting of two ClO3 portions linked by an oxygen atom. It has an overall bent molecular geometry (C2 symmetry), with a Cl−O−Cl angle of 118.6°. The chlorine–oxygen bond lengths are 1.709 Å in the central region and 1.405 Å within each ClO3 cluster.[1] In this compound, chlorine exists in its highest formal oxidation state of +7.
Chemistry
editDichlorine heptoxide reacts with primary and secondary amines in carbon tetrachloride solution to yield perchloric amides:[4]
- 2 RNH
2 + Cl
2O
7 → 2 RNHClO
3 + H
2O - 2 R
2NH + Cl
2O
7 → 2 R
2NClO
3 + H
2O
It also reacts with alkenes to give alkyl perchlorates. For example, it reacts with propene in carbon tetrachloride solution to yield isopropyl perchlorate and 1-chloro-2-propyl perchlorate.[5]
Dichlorine heptoxide reacts with alcohols to form alkyl perchlorates.[6]
Dichlorine heptoxide is a strongly acidic oxide, and in solution it forms an equilibrium with perchloric acid.
Safety
editAlthough it is the most stable chlorine oxide, Cl2O7 is a strong oxidizer as well as an explosive that can be set off with flame or mechanical shock, or by contact with iodine.[7] Nevertheless, it is less strongly oxidising than the other chlorine oxides, and does not attack sulfur, phosphorus, or paper when cold.[1] It has the same effects on the human body as elemental chlorine, and requires the same precautions.[8]
References
edit- ^ a b c d Holleman, Arnold F.; Wiberg, Egon (2001). Inorganic chemistry. Translated by Mary Eagleson; William Brewer. San Diego: Academic Press. p. 464. ISBN 0-12-352651-5.
- ^ Byrns, A. C.; Rollefson, G. K. (1934). "The Formation of Chlorine Heptoxide on Illumination of Mixtures of Chlorine and Ozone". Journal of the American Chemical Society. 56 (5): 1250–1251. doi:10.1021/ja01320a506.
- ^ Martin, Jan M. L (2006-10-12). "Heats of formation of perchloric acid, HClO4, and perchloric anhydride, Cl2O7. Probing the limits of W1 and W2 theory". Journal of Molecular Structure: THEOCHEM. Modelling Structure and Reactivity: the 7th triennial conference of the World Association of Theoritical and Computational Chemists (WATOC 2005). 771 (1): 19–26. arXiv:physics/0508076. doi:10.1016/j.theochem.2006.03.035. ISSN 0166-1280. S2CID 98385268.
- ^ Beard, C. D.; Baum, K. (1974). "Reactions of dichlorine heptoxide with amines". Journal of the American Chemical Society. 96 (10): 3237–3239. doi:10.1021/ja00817a034.
- ^ Baum, K. . (1976). "Reactions of dichlorine heptoxide with olefins". The Journal of Organic Chemistry. 41 (9): 1663–1665. doi:10.1021/jo00871a048.
- ^ Baum, Kurt; Beard, Charles D. (1974-05-01). "Reactions of dichlorine heptoxide with alcohols". Journal of the American Chemical Society. 96 (10): 3233–3237. doi:10.1021/ja00817a033. ISSN 0002-7863.
- ^ Lewis, Robert Alan (1998). Lewis' dictionary of toxicology. CRC Press. p. 260. ISBN 1-56670-223-2.
- ^ Jeanne Mager Stellman, ed. (1998). "Halogens and their compounds". Encyclopaedia of occupational health and safety (4th ed.). International Labour Organization. p. 104.210. ISBN 92-2-109817-6.
