Dopamine, sold under the brand name Intropin among others, is a medication most commonly used in the treatment of very low blood pressure, a slow heart rate that is causing symptoms, and, if epinephrine is not available, cardiac arrest.[4] In newborn babies it continues to be the preferred treatment for very low blood pressure.[5] In children epinephrine or norepinephrine is generally preferred while in adults norepinephrine is generally preferred for very low blood pressure.[6][7] It is given intravenously or intraosseously as a continuous infusion.[4] Effects typically begin within five minutes.[4] Doses are then increased to effect.[4]
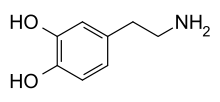 Skeletal formula of dopamine | |
 | |
| Clinical data | |
|---|---|
| Trade names | Intropin, Dopastat, Revimine, others |
| Other names | 2-(3,4-Dihydroxyphenyl)ethylamine; 3,4-Dihydroxyphenethylamine; 3-hydroxytyramine; DA; Intropin; Revivan; Oxytyramine; Prolactin inhibiting factor; Prolactin inhibiting hormone |
| AHFS/Drugs.com | Monograph |
| Routes of administration | Intravenous |
| ATC code | |
| Physiological data | |
| Source tissues | Substantia nigra; ventral tegmental area; many others |
| Target tissues | System-wide |
| Receptors | D1, D2, D3, D4, D5, TAAR1[2] |
| Agonists | Direct: apomorphine, bromocriptine Indirect: cocaine, substituted amphetamine, cathinone, bupropion |
| Antagonists | Neuroleptics, metoclopramide, domperidone |
| Metabolism | MAO, COMT,[2] ALDH, DBH, MAO-A, MAO-B, COMT |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Metabolism | MAO, COMT,[2] ALDH, DBH, MAO-A, MAO-B, COMT |
| Excretion | Kidney |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C8H11NO2 |
| Molar mass | 153.181 g·mol−1 |
| 3D model (JSmol) | |
| Density | 1.26 g/cm3 |
| Melting point | 128 °C (262 °F) |
| Boiling point | decomposes |
| |
| |
| (verify) | |
Common side effects include worsening kidney function, an irregular heartbeat, chest pain, vomiting, headache, or anxiety.[4] If it enters into the soft tissue around the vein local tissue death may occur.[4] The medication phentolamine can be given to try to decrease this risk.[4] It is unclear if dopamine is safe to use during pregnancy or breastfeeding.[4] At low doses dopamine mainly triggers dopamine receptors and β1-adrenergic receptors while at high doses it works via α-adrenergic receptors.[4]
Dopamine was first synthesized in a laboratory in 1910 by George Barger and James Ewens in England.[8] It is on the World Health Organization's List of Essential Medicines.[9] In human physiology dopamine is a neurotransmitter as well as a hormone.[10]
Medical uses
editLow blood pressure
editIn newborn babies it continues to be the preferred treatment for very low blood pressure.[5] In children epinephrine or norepinephrine is generally preferred while in adults norepinephrine is generally preferred for very low blood pressure.[6][7]
In those with low blood volume or septic shock, this should be corrected with intravenous fluids before dopamine is considered.[4]
Kidney function
editLow-dosage dopamine has been routinely used for the treatment and prevention of acute kidney injury. However, since 1999 a number of reviews have concluded that doses at such low levels are not effective and may sometimes be harmful.[11][12]
Administration
editSince the half-life of dopamine in plasma is short—approximately one minute in adults, two minutes in newborn babies and up to five minutes in preterm babies—it is usually given as a continuous intravenous drip rather than a single injection.[13]
Other
editA fluorinated form of L-DOPA known as fluorodopa is available for use in positron emission tomography to assess the function of the nigrostriatal pathway.[14]
Contraindications
editDopamine should generally not be given to people who have a pheochromocytoma or uncorrected very fast heart rate.[4]
Side effects
editThe LD50, or dose which is expected to prove lethal in 50% of the population, has been found to be: 59 mg/kg (mouse; administered intravenously); 950 mg/kg (mouse; administered intraperitoneally); 163 mg/kg (rat; administered intraperitoneally); 79 mg/kg (dog; administered intravenously).[15]
Extravasation
editIf extravasation occurs local tissue death may result.[4] The medication phentolamine can be injected at the site to try to decrease the risk of tissue death.[4]
Mechanism of action
editIts effects, depending on dosage, include an increase in sodium excretion by the kidneys, an increase in urine output, an increase in heart rate, and an increase in blood pressure.[13] At low doses it acts through the sympathetic nervous system to increase heart muscle contraction force and heart rate, thereby increasing cardiac output and blood pressure.[16] Higher doses also cause vasoconstriction that further increases blood pressure.[16][17]
While some effects result from stimulation of dopamine receptors, the prominent cardiovascular effects result from dopamine acting at α1, β1, and β2 adrenergic receptors.[18][19]
Society and culture
editLegal status
editIn March 2024, the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (EMA) adopted a positive opinion, recommending the granting of a pediatric use marketing authorization (PUMA) for the medicinal product Neoatricon, intended for treatment of hypotension in neonates, infants and children under 18 years of age.[3][20] The applicant for this medicinal product is BrePco Biopharma Limited.[3]
References
edit- ^ Cruickshank L, Kennedy AR, Shankland N (2013). "Tautomeric and ionisation forms of dopamine and tyramine in the solid state". J. Mol. Struct. 1051: 132–136. Bibcode:2013JMoSt1051..132C. doi:10.1016/j.molstruc.2013.08.002.
- ^ a b "Dopamine: Biological activity". IUPHAR/BPS guide to pharmacology. International Union of Basic and Clinical Pharmacology. Archived from the original on 5 February 2016. Retrieved 29 January 2016.
- ^ a b c "Neoatricon EPAR". European Medicines Agency (EMA). 21 March 2024. Retrieved 23 March 2024. Text was copied from this source which is copyright European Medicines Agency. Reproduction is authorized provided the source is acknowledged.
- ^ a b c d e f g h i j k l m "Dopamine Hydrochloride". drugs.com. American Society of Health-System Pharmacists. 29 June 2016. Archived from the original on 14 September 2016. Retrieved 15 July 2016.
- ^ a b Bhayat SI, Gowda HM, Eisenhut M (May 2016). "Should dopamine be the first line inotrope in the treatment of neonatal hypotension? Review of the evidence". World Journal of Clinical Pediatrics. 5 (2): 212–222. doi:10.5409/wjcp.v5.i2.212. PMC 4857235. PMID 27170932.
- ^ a b De Backer D, Aldecoa C, Njimi H, Vincent JL (March 2012). "Dopamine versus norepinephrine in the treatment of septic shock: a meta-analysis*". Critical Care Medicine. 40 (3): 725–730. doi:10.1097/ccm.0b013e31823778ee. PMID 22036860. S2CID 24620964.
- ^ a b Dellinger RP, Levy MM, Rhodes A, Annane D, Gerlach H, Opal SM, et al. (February 2013). "Surviving sepsis campaign: international guidelines for management of severe sepsis and septic shock: 2012". Critical Care Medicine. 41 (2): 580–637. doi:10.1097/CCM.0b013e31827e83af. PMID 23353941. S2CID 34855187.
- ^ Fahn S (2008). "The history of dopamine and levodopa in the treatment of Parkinson's disease". Movement Disorders. 23 (Suppl 3): S497–S508. doi:10.1002/mds.22028. PMID 18781671. S2CID 45572523.
According to Hornykiewicz,6 dopamine was first synthesized by George Barger and James Ewens in 1910 at the Wellcome labs in London, England.
- ^ World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ^ Millar T (2002). Biochemistry explained : a practical guide to learning biochemistry. London: Routledge. p. 40. ISBN 9780415299411. Archived from the original on 15 August 2016.
- ^ Karthik S, Lisbon A (2006). "Low-dose dopamine in the intensive care unit". Seminars in Dialysis. 19 (6): 465–471. doi:10.1111/j.1525-139X.2006.00208.x. PMID 17150046. S2CID 22538344.
- ^ Power DA, Duggan J, Brady HR (April 1999). "Renal-dose (low-dose) dopamine for the treatment of sepsis-related and other forms of acute renal failure: ineffective and probably dangerous". Clinical and Experimental Pharmacology & Physiology. Supplement. 26: S23–S28. PMID 10386250.
- ^ a b Bhatt-Mehta V, Nahata MC (1989). "Dopamine and dobutamine in pediatric therapy". Pharmacotherapy. 9 (5): 303–314. doi:10.1002/j.1875-9114.1989.tb04142.x. PMID 2682552. S2CID 25614283.
- ^ Deng WP, Wong KA, Kirk KL (2002). "Convenient syntheses of 2-, 5- and 6-fluoro- and 2,6-difluoro-L-DOPA". Tetrahedron: Asymmetry. 13 (11): 1135–1140. doi:10.1016/S0957-4166(02)00321-X.
- ^ Lewis RJ (2004). Sax's Dangerous Properties of Industrial Materials, 11th Ed. Hoboken, NJ.: Wiley & Sons. p. 1552. ISBN 978-0-471-47662-7.
- ^ a b Bronwen JB, Knights KM (2009). Pharmacology for Health Professionals (2nd ed.). Elsevier Australia. p. 192. ISBN 978-0-7295-3929-6.
- ^ De Backer D, Biston P, Devriendt J, Madl C, Chochrad D, Aldecoa C, et al. (March 2010). "Comparison of dopamine and norepinephrine in the treatment of shock". The New England Journal of Medicine. 362 (9): 779–789. doi:10.1056/NEJMoa0907118. PMID 20200382. S2CID 2208904.
- ^ Moses S. "Dopamine". Family Practice Notebook. Archived from the original on 1 February 2016. Retrieved 1 February 2016.
Dopamine binds to alpha-1 and beta-1 adrenergic receptors. Mediated through myocardial beta-1 adrenergic receptors, dopamine increase heart rate and force, thereby increasing cardiac output. Alpha-1 adrenergic receptor stimulation on vascular smooth muscle, leads to vasoconstriction and results in an increase in systemic vascular resistance
- ^ Katritsis DG, Gersh BJ, Camm AJ (19 September 2013). Clinical Cardiology: Current Practice Guidelines. OUP Oxford. p. 314. ISBN 9780191508516. Archived from the original on 6 May 2016.
Dopamine binds to beta-1, beta-2, alpha-1 and dopaminergic receptors.
- ^ "Meeting highlights from the Committee for Medicinal Products for Human Use (CHMP) 18-21 March 2024". European Medicines Agency (Press release). 22 March 2024. Retrieved 13 June 2024.