The Horner–Wadsworth–Emmons (HWE) reaction is a chemical reaction used in organic chemistry of stabilized phosphonate carbanions with aldehydes (or ketones) to produce predominantly E-alkenes.[1]
| Horner–Wadsworth–Emmons reaction | |
|---|---|
| Named after | Leopold Horner William S. Wadsworth William D. Emmons |
| Reaction type | Coupling reaction |
| Identifiers | |
| Organic Chemistry Portal | wittig-horner-reaction |
| RSC ontology ID | RXNO:0000056 |
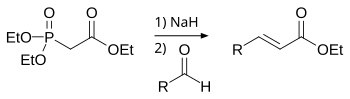
In 1958, Leopold Horner published a modified Wittig reaction using phosphonate-stabilized carbanions.[2][3] William S. Wadsworth and William D. Emmons further defined the reaction.[4][5]
In contrast to phosphonium ylides used in the Wittig reaction, phosphonate-stabilized carbanions are more nucleophilic but less basic. Likewise, phosphonate-stabilized carbanions can be alkylated. Unlike phosphonium ylides, the dialkylphosphate salt byproduct is easily removed by aqueous extraction.
Reaction mechanism
editThe Horner–Wadsworth–Emmons reaction begins with the deprotonation of the phosphonate to give the phosphonate carbanion 1. Nucleophilic addition of the carbanion onto the aldehyde 2 (or ketone) producing 3a or 3b is the rate-limiting step.[12] If R2 = H, then intermediates 3a and 4a and intermediates 3b and 4b can interconvert with each other.[13] The final elimination of oxaphosphetanes 4a and 4b yield (E)-alkene 5 and (Z)-alkene 6, with the by-product being a dialkyl-phosphate.
The ratio of alkene isomers 5 and 6 is not dependent upon the stereochemical outcome of the initial carbanion addition and upon the ability of the intermediates to equilibrate.
The electron-withdrawing group (EWG) alpha to the phosphonate is necessary for the final elimination to occur. In the absence of an electron-withdrawing group, the final product is the β-hydroxyphosphonate 3a and 3b.[14] However, these β-hydroxyphosphonates can be transformed to alkenes by reaction with diisopropylcarbodiimide.[15]
Stereoselectivity
editThe Horner–Wadsworth–Emmons reaction favours the formation of (E)-alkenes. In general, the more equilibration amongst intermediates, the higher the selectivity for (E)-alkene formation.
Disubstituted alkenes
editThompson and Heathcock have performed a systematic study of the reaction of methyl 2-(dimethoxyphosphoryl)acetate with various aldehydes.[16] While each effect was small, they had a cumulative effect making it possible to modify the stereochemical outcome without modifying the structure of the phosphonate. They found greater (E)-stereoselectivity with the following conditions:
- Increasing steric bulk of the aldehyde
- Higher reaction temperatures (23 °C over −78 °C)
- Li > Na > K salts
In a separate study, it was found that bulky phosphonate and bulky electron-withdrawing groups enhance E-alkene selectivity.
Trisubstituted alkenes
editThe steric bulk of the phosphonate and electron-withdrawing groups plays a critical role in the reaction of α-branched phosphonates with aliphatic aldehydes.[17]
| R1 | R2 | Ratio of alkenes ( E : Z ) |
|---|---|---|
| Methyl | Methyl | 5 : 95 |
| Methyl | Ethyl | 10 : 90 |
| Ethyl | Ethyl | 40 : 60 |
| Isopropyl | Ethyl | 90 : 10 |
| Isopropyl | Isopropyl | 95 : 5 |
Aromatic aldehydes produce almost exclusively (E)-alkenes. In case (Z)-alkenes from aromatic aldehydes are needed, the Still–Gennari modification (see below) can be used.
Olefination of ketones
editThe stereoselectivity of the Horner–Wadsworth–Emmons reaction of ketones is poor to modest.
Variations
editBase sensitive substrates
editSince many substrates are not stable to sodium hydride, several procedures have been developed using milder bases. Masamune and Roush have developed mild conditions using lithium chloride and DBU.[18] Rathke extended this to lithium or magnesium halides with triethylamine.[19] Several other bases have been found effective.[20][21][22]
Gennari-Still modification
editW. Clark Still and C. Gennari have developed conditions that give Z-alkenes with excellent stereoselectivity.[23][24] Using phosphonates with electron-withdrawing groups (trifluoroethyl[25]) together with strongly dissociating conditions (KHMDS and 18-crown-6 in THF) nearly exclusive Z-alkene production can be achieved.
Ando has suggested that the use of electron-deficient phosphonates accelerates the elimination of the oxaphosphetane intermediates.[26]
See also
editReferences
edit- ^ Wadsworth, W. Org. React. 1977, 25, 73. doi:10.1002/0471264180.or025.02
- ^ Leopold Horner; Hoffmann, H. M. R.; Wippel, H. G. Ber. 1958, 91, 61–63.
- ^ Horner, L.; Hoffmann, H. M. R.; Wippel, H. G.; Klahre, G. Ber. 1959, 92, 2499–2505.
- ^ Wadsworth, W. S., Jr.; Emmons, W. D. J. Am. Chem. Soc. 1961, 83, 1733. (doi:10.1021/ja01468a042)
- ^ Wadsworth, W. S., Jr.; Emmons, W. D. Organic Syntheses, Coll. Vol. 5, p. 547 (1973); Vol. 45, p. 44 (1965). (Article)
- ^ Wadsworth, W. S., Jr. Org. React. 1977, 25, 73–253. (Review)
- ^ Boutagy, J.; Thomas, R. Chem. Rev. 1974, 74, 87–99. (Review, doi:10.1021/cr60287a005)
- ^ Kelly, S. E. Compr. Org. Synth. 1991, 1, 729–817. (Review)
- ^ B. E. Maryanoff; Reitz, A. B. Chem. Rev. 1989, 89, 863–927. (Review, doi:10.1021/cr00094a007)
- ^ Bisceglia, J. A., Orelli, L. R. Curr. Org. Chem. 2012, 16, 2206–2230 (Review)
- ^ Bisceglia, J. A., Orelli, L. R. Curr. Org. Chem. 2015, 19, 744–775 (Review)
- ^ Larsen, R. O.; Aksnes, G. Phosphorus Sulfur 1983, 15, 218–219.
- ^ Lefèbvre, G.; Seyden-Penne, J. J. Chem Soc., Chem. Commun. 1970, 1308–09.
- ^ Corey, E. J.; Kwiatkowski, G. T. J. Am. Chem. Soc. 1966, 88, 5654–56. (doi:10.1021/ja00975a057)
- ^ Reichwein, J. F.; Pagenkopf, B. L. J. Am. Chem. Soc. 2003, 125, 1821–24. (doi:10.1021/ja027658s)
- ^ Thompson, S. K.; Heathcock, C. H. J. Org. Chem. 1990, 55, 3386–88. (doi:10.1021/jo00297a076)
- ^ Nagaoka, H.; Kishi, Y. Tetrahedron 1981, 37, 3873–3888.
- ^ Blanchette, M. A.; Choy, W.; Davis, J. T.; Essenfeld, A. P.; Masamune, S.; Roush, W. R.; Sakai, T. Tetrahedron Letters 1984, 25, 2183–2186.
- ^ Rathke, M. W.; Nowak, M. J. Org. Chem. 1985, 50, 2624–2626. (doi:10.1021/jo00215a004)
- ^ Paterson, I.; Yeung, K.-S.; Smaill, J. B. Synlett 1993, 774.
- ^ Simoni, D.; Rossi, M.; Rondanin, R.; Mazzali, A.; Baruchello, R.; Malagutti, C.; Roberti, M.; Invidiata, F. P. Org. Letters 2000, 2, 3765–3768.
- ^ Blasdel, L. K.; Myers, A. G. Org. Letters 2005, 7, 4281–4283.
- ^ Still, W. C.; Gennari, C. Tetrahedron Letters 1983, 24, 4405–4408.
- ^ Janicki, Ignacy; Kiełbasiński, Piotr (2020). "Still–Gennari Olefination and its Applications in Organic Synthesis". Advanced Synthesis & Catalysis. 362 (13): 2552–2596. doi:10.1002/adsc.201901591. ISSN 1615-4169. S2CID 216228029.
- ^ Patois, C.; Savignac, P.; About-Jaudet, E.; Collignon, N. Organic Syntheses, Coll. Vol. 9, p. 88 (1998); Vol. 73, p. 152 (1996). (Article)
- ^ Ando, K. J. Org. Chem. 1997, 62, 1934–1939. (doi:10.1021/jo970057c)