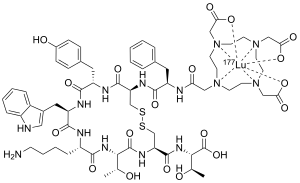
Lutetium (177Lu) oxodotreotide (INN) or 177Lu dotatate, brand name Lutathera, is a chelated complex of a radioisotope of the element lutetium with dotatate, used in peptide receptor radionuclide therapy. Specifically, it is used in the treatment of cancers which express somatostatin receptors.[5] It is a radiolabeled somatostatin analog.[3][6][7]
 | |
| Clinical data | |
|---|---|
| Trade names | Lutathera |
| AHFS/Drugs.com | Monograph |
| License data | |
| Routes of administration | Intravenous |
| Drug class | Radiolabeled somatostatin analog |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| UNII | |
| KEGG | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C65H87LuN14O19S2 |
| Molar mass | 1607.58 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Alternatives to 177Lu-dotatate include yttrium-90 dotatate or DOTATOC. The longer range of the beta particles emitted by 90Y, which deliver the therapeutic effect, may make it more suitable for large tumors with 177Lu reserved for smaller volumes[8][9]
The US Food and Drug Administration (FDA) considers 177Lu dotatate to be a first-in-class medication.[10]
Medical uses
editIn the US, 177Lu dotatate is indicated for the treatment of somatostatin receptor-positive gastroenteropancreatic neuroendocrine tumors (GEP-NETs), including foregut, midgut, and hindgut neuroendocrine tumors in adults.[3][6][7]
In the EU, lutetium (177Lu) oxodotreotide is indicated for the treatment of unresectable or metastatic, progressive, well differentiated (G1 and G2), somatostatin receptor positive gastroenteropancreatic neuroendocrine tumours (GEP-NETs) in adults.[4]
Adverse effects
editThe therapeutic effect of 177Lu derives from the ionizing beta radiation it emits, however this can also be harmful to healthy tissue and organs. The kidneys are particularly at risk as they help to remove 177Lu dotatate from the body.[11] To protect them, an amino acid solution (arginine/lysine) is administered by slow infusion, starting before the radioactive administration and normally continuing for several hours afterwards.[9][12][13]
History
editThe European Commission approved lutetium (177Lu) oxodotreotide (brand name Lutathera) "for the treatment of unresectable or metastatic, progressive, well differentiated (G1 and G2), somatostatin receptor positive gastroenteropancreatic neuroendocrine tumours (GEP-NETs) in adults" in September 2017.[14][4]
177Lu dotatate was approved in the United States for the treatment of SSTR positive gastroenteropancreatic neuroendocrine tumors (GEP-NETs), including foregut, midgut and hindgut neuroendocrine tumors in adults, in January 2018.[3][6][7] This was the first time a radiopharmaceutical had been approved for the treatment of GEP-NETs in the United States.[6]
The US Food and Drug Administration (FDA) approved 177Lu dotatate based primarily on evidence from one clinical trial, NETTER-1 of 229 participants with somatostatin-receptor positive midgut GEP-NETs.[15] Enrolled participants had tumors which could not be surgically removed and were worsening while receiving treatment with octreotide.[15]
Participants were randomly assigned to receive either 177Lu dotatate with long-acting octreotide or long-acting octreotide, at a higher dose, alone.[15] 177Lu dotatate was injected through the vein and long-acting octreotide was injected in the muscle.[15] Both, participants and health care providers knew which treatment was given.[15] The benefit of 177Lu dotatate was evaluated by measuring the length of time that tumors did not grow after treatment and compared it to the control group (progression free survival).[15]
The FDA considered additional data from a second study based on data from 1,214 participants with somatostatin receptor-positive tumors, including GEP-NETS, who received 177Lu dotatate at a single site in the Netherlands, Erasmus MC.[6][15] All participants received 177Lu dotatate with octreotide.[15] Participants and health care providers knew which treatment was given.[15] The benefit of 177Lu dotatate was evaluated by measuring if and how much the tumor size changed during treatment (the overall response rate).[15] Complete or partial tumor shrinkage was reported in 16 percent of a subset of 360 participants with GEP-NETs who were evaluated for response by the FDA.[6] Participants initially enrolled in the study received 177Lu dotatate as part of an expanded access program.[6]
The FDA granted the application for 177Lu dotatate priority review and orphan drug designations.[6] The FDA granted the approval of Lutathera to Advanced Accelerator Applications.[6]
In April 2024, the FDA approved 177Lu dotatate for the treatment of children aged 12 years and older with somatostatin receptor-positive (SSTR)-positive gastroenteropancreatic neuroendocrine tumors (GEP-NETs), including foregut, midgut, and hindgut neuroendocrine tumors.[16][17] It was approved for adults in 2018.[16] This is the first FDA approval of a radioactive drug, or radiopharmaceutical, for children aged twelve years of age and older with SSTR-positive gastroenteropancreatic neuroendocrine tumors.[16]
Approval for children aged 12 years and older was based on pharmacokinetic, dosimetry, and safety data from NETTER-P (NCT04711135), an ongoing, international, multi-center, open-label, single-arm study of lutetium Lu 177 dotatate in adolescents with locally advanced/inoperable or metastatic SSTR-positive gastroenteropancreatic neuroendocrine tumors or pheochromocytoma/paraganglioma.[16] Approval was also based on the extrapolation of efficacy outcomes observed in NETTER-1 (NCT01578239), a randomized, multicenter, open-label, active-controlled trial in 229 participants with locally advanced/inoperable or metastatic SSTR-positive midgut carcinoid tumors, which supported the original approval of lutetium Lu 177 dotatate in adults.[16]
Safety was evaluated in nine pediatric participants in NETTER-P, including four participants with gastroenteropancreatic neuroendocrine tumors.[16] The major outcome measures were absorbed radiation doses in target organs and incidence of adverse reactions after the first treatment cycle.[16] Additional outcome measures included short-term adverse reactions following treatment with lutetium Lu 177 dotatate.[16] The adverse reaction profile observed in NETTER-P was similar to that observed in adults.[16]
References
edit- ^ "Summary Basis of Decision (SBD) for Lutathera". Health Canada. 23 October 2014. Archived from the original on 31 May 2022. Retrieved 29 May 2022.
- ^ "Lutathera 370 MBq/mL solution for infusion - Summary of Product Characteristics (SmPC)". (emc). Archived from the original on 9 July 2021. Retrieved 9 July 2021.
- ^ a b c d "Lutathera- lutetium lu 177 dotatate injection". DailyMed. 4 May 2020. Archived from the original on 16 November 2020. Retrieved 8 November 2020.
- ^ a b c "Lutathera EPAR". European Medicines Agency (EMA). 17 September 2018. Archived from the original on 11 December 2019. Retrieved 11 December 2019.
- ^ Wang L, Tang K, Zhang Q, Li H, Wen Z, Zhang H, et al. (2013). "Somatostatin receptor-based molecular imaging and therapy for neuroendocrine tumors". BioMed Research International. 2013: 102819. doi:10.1155/2013/102819. PMC 3784148. PMID 24106690.
- ^ a b c d e f g h i "FDA approves new treatment for certain digestive tract cancers". U.S. Food and Drug Administration (FDA) (Press release). 26 January 2018. Archived from the original on 11 December 2019. Retrieved 11 December 2019. This article incorporates text from this source, which is in the public domain.
- ^ a b c "FDA approves lutetium Lu 177 dotatate for treatment of GEP-NETS". U.S. Food and Drug Administration (FDA) (Press release). 26 January 2018. Archived from the original on 11 December 2019. Retrieved 11 December 2019. This article incorporates text from this source, which is in the public domain.
- ^ Ramage JK, Ahmed A, Ardill J, Bax N, Breen DJ, Caplin ME, et al. (January 2012). "Guidelines for the management of gastroenteropancreatic neuroendocrine (including carcinoid) tumours (NETs)". Gut. 61 (1): 6–32. doi:10.1136/gutjnl-2011-300831. PMC 3280861. PMID 22052063.
- ^ a b Bodei L, Mueller-Brand J, Baum RP, Pavel ME, Hörsch D, O'Dorisio MS, et al. (May 2013). "The joint IAEA, EANM, and SNMMI practical guidance on peptide receptor radionuclide therapy (PRRNT) in neuroendocrine tumours". European Journal of Nuclear Medicine and Molecular Imaging. 40 (5): 800–16. doi:10.1007/s00259-012-2330-6. PMC 3622744. PMID 23389427.
- ^ New Drug Therapy Approvals 2018 (PDF). U.S. Food and Drug Administration (FDA) (Report). January 2019. Archived from the original on 17 September 2020. Retrieved 16 September 2020.
- ^ Thompson L (7 February 2019). "Significance of Amino Acid Solution With Lutetium Lu 177 Dotatate". Oncology Nurse Advisor. Archived from the original on 17 October 2021. Retrieved 2 November 2020.
- ^ "LysaKare EPAR". European Medicines Agency (EMA). 24 May 2019. Archived from the original on 23 July 2020. Retrieved 22 July 2020.
- ^ Volterrani D, Erba PA, Carrió I, Strauss HW, Mariani G (10 August 2019). Nuclear Medicine Textbook: Methodology and Clinical Applications. Springer. p. 782. ISBN 978-3-319-95564-3. Archived from the original on 10 January 2023. Retrieved 7 November 2020.
- ^ "European approval of lutetium oxodotreotide for gastroenteropancreatic neuroendocrine (GEP-NET) tumours". ecancer.org. 3 October 2017. Archived from the original on 3 April 2018. Retrieved 2 April 2018.
- ^ a b c d e f g h i j "Drug Trials Snapshots: Lutathera". U.S. Food and Drug Administration (FDA). 20 February 2018. Archived from the original on 11 December 2019. Retrieved 11 December 2019. This article incorporates text from this source, which is in the public domain.
- ^ a b c d e f g h i "FDA approves lutetium Lu 177 dotatate for pediatric patients 12 years". U.S. Food and Drug Administration. 23 April 2024. Archived from the original on 25 April 2024. Retrieved 25 April 2024. This article incorporates text from this source, which is in the public domain.
- ^ "Novartis Gets FDA Approval for Lutathera in Pediatric Treatment". MarketScreener. 23 April 2024. Archived from the original on 23 April 2024. Retrieved 23 April 2024.