Mannose-binding lectin (MBL), also called mannan-binding lectin or mannan-binding protein (MBP), is a lectin that is instrumental in innate immunity[5][6] as an opsonin and via the lectin pathway.
Structure
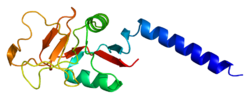
editMBL has an oligomeric structure (400-700 kDa), built of subunits that contain three presumably identical peptide chains of about 30 kDa each.
Although MBL can form several oligomeric forms, there are indications that dimers and trimers are biologically inactive as an opsonin and at least a tetramer form is needed for activation of complement.[7]
Genes and polymorphisms
editHuman MBL2 gene is located on chromosome 10q11.2-q21.[8] Mice have two homologous genes, but in human the first of them was lost. A low level expression of an MBL1 pseudogene 1 (MBL1P1) was detected in liver. The pseudogene encodes a truncated 51-amino acid protein that is homologous to the MBLA isoform in rodents and some primates.[9]
Structural mutations in exon 1 of the human MBL2 gene, at codon 52 (Arg to Cys, allele D), codon 54 (Gly to Asp, allele B) and codon 57 (Gly to Glu, allele C), also independently reduce the level of functional serum MBL by disrupting the collagenous structure of the protein.[10] Furthermore, several nucleotide substitutions in the promoter region of the MBL2 gene at position −550 (H/L polymorphism), −221 (X/Y polymorphism) and −427, −349, −336, del (−324 to −329), −70 and +4 (P/Q polymorphisms) affect the MBL serum concentration. Both the frequency of structural mutations and the promoter polymorphisms that are in strong linkage disequilibrium vary among ethnic groups resulting in seven major haplotypes: HYPA, LYQA, LYPA, LXPA, LYPB, LYQC and HYPD. Differences in the distribution of these haplotypes are the major cause of interracial variations in MBL serum levels. Both HYPA and LYQA are high-producing haplotypes, LYPA intermediate-producing haplotype and LXPA low-producing haplotype, whereas LYPB, LYQC and HYPD are defective haplotypes, which cause a severe MBL deficiency.[11] Such polymorphism also present in exon 4.[12]
Both MBL2 and MBL1P1 genes has been repeatedly hit throughout evolution of primates. The latter silenced eventually by mutations in the glycine residues of the collagen-like region. It has been selectively turned off during evolution through the same molecular mechanisms causing the MBL2 variant alleles in man, suggesting an evolutionary selection for low-producing MBL genes.[10]
Posttranslational modifications
editIn rat hepatocytes, MBL is synthesized in the rough endoplasmic reticulum. While in Golgi, it undergoes two distinct posttranslational modifications and is assembled into high molecular weight multimeric complexes. The modifications produce MBL in multiple forms of slightly various molecular masses and pI from 5.7 to 6.2.[13] Proteolytic cleavage also resulted in removal of the 20-aa N-terminal signal peptide,[14] and hydroxylation and glycosylation were also detected.[13] Some cysteine residues can be converted to dehydroalanin.[15]
Function
editMBL belongs to the class of collectins in the C-type lectin superfamily, whose function appears to be pattern recognition in the first line of defense in the pre-immune host. MBL recognizes carbohydrate patterns found on the surface of a large number of pathogenic micro-organisms, including bacteria, viruses, protozoa and fungi. Binding of MBL to a micro-organism results in activation of the lectin pathway of the complement system.
Another important function of MBL is that this molecule binds senescent[16] and apoptotic cells and enhances engulfment of whole, intact apoptotic cells, as well as cell debris by phagocytes.[17][18]
Activation
editThe complement system can be activated through three pathways: the classical pathway, the alternative pathway, and the lectin pathway. One way the most-recently discovered lectin pathway is activated is through mannose-binding lectin protein. MBL binds to carbohydrates (to be specific, D-mannose and L-fucose residues) found on the surfaces of many pathogens.
For example, MBL has been shown to bind to:
- yeasts such as Candida albicans[19][20]
- viruses such as HIV[21] and influenza A
- many bacteria, including Salmonella and Streptococci
- parasites like Leishmania
- SARS-CoV-2[22][23]
Complexes
editMBL in the blood is complexed with (bound to) a serine protease called MASP (MBL-associated serine protease). There are three MASPs: MASP-1, MASP-2 and MASP-3, which have protease domains. There are also sMAP (also called MAp19) and MAp44, which do not have protease domains and are thought to be regulatory molecules of MASPs. MASPs also form complexes with ficolins, which are similar to MBL functionally and structurally with the exception that ficolins recognize their targets through fibrinogen-like domains, unlike MBL.
In order to activate the complement system when MBL binds to its target (for example, mannose on the surface of a bacterium), the MASP protein functions to cleave the blood protein C4 into C4a and C4b. The C4b fragments can then bind to the surface of the bacterium, and initiate the formation of a C3-convertase.
The subsequent complement cascade catalyzed by C3-convertase results in creating a membrane attack complex, which causes lysis of the pathogen as well as altered-self in the context of apoptotic and necrotic cells.
MBL/MASP-1 complex also has thrombin-like activity (thrombin clots fibrin to initiate blood clots). Mice that genetically lack MBL or MASP-1/3 (but not MASP-2/sMAP) have prolonged bleeding time in experimental injury models, although mice are seen to be normal if there is no insult to the body.
Clinical significance
editIt is produced in the liver as a response to infection, and is part of many other factors termed acute phase proteins.[24] Expression and function in other organs were also suggested.[25] The three structural polymorphisms of exon 1 have been reported to cause susceptibility to various common infections, including meningococcal disease.[26][27] However, evidence has been presented that suggests no harmful effect of these variants with regard to mengingococcal disease.[28] MBL deficiency is very common in humans, with approximately 10% of individuals having this deficiency.[29]
External links
edit- Mannan-Binding+Lectin at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
References
edit- ^ a b c GRCh38: Ensembl release 89: ENSG00000165471 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000024863 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ Fraser IP, Koziel H, Ezekowitz RA (1998). "The serum mannose-binding protein and the macrophage mannose receptor are pattern recognition molecules that link innate and adaptive immunity". Semin. Immunol. 10 (5): 363–72. doi:10.1006/smim.1998.0141. PMID 9799711.
- ^ Worthley DL, Bardy PG, Mullighan CG (2005). "Mannose-binding lectin: biology and clinical implications". Internal Medicine Journal. 35 (9): 548–55. doi:10.1111/j.1445-5994.2005.00908.x. PMID 16105157. S2CID 40041919.
- ^ Sheriff S, Chang CY, Ezekowitz RA (November 1994). "Human mannose-binding protein carbohydrate recognition domain trimerizes through a triple alpha-helical coiled-coil". Nat. Struct. Biol. 1 (11): 789–94. doi:10.1038/nsb1194-789. PMID 7634089. S2CID 5871944.
- ^ Sastry K, Herman GA, Day L, Deignan E, Bruns G, Morton CC, Ezekowitz RA (October 1989). "The human mannose-binding protein gene. Exon structure reveals its evolutionary relationship to a human pulmonary surfactant gene and localization to chromosome 10". J. Exp. Med. 170 (4): 1175–89. doi:10.1084/jem.170.4.1175. PMC 2189467. PMID 2477486.
- ^ Guo N, Mogues T, Weremowicz S, Morton CC, Sastry KN (March 1998). "The human ortholog of rhesus mannose-binding protein-A gene is an expressed pseudogene that localizes to chromosome 10". Mamm. Genome. 9 (3): 246–9. doi:10.1007/s003359900735. PMID 9501312. S2CID 12065976.
- ^ a b Seyfarth J, Garred P, Madsen HO (2005). "The 'involution' of mannose-binding lectin". Human Molecular Genetics. 14 (19): 2859–69. doi:10.1093/hmg/ddi318. PMID 16115813.
- ^ Online Mendelian Inheritance in Man (OMIM): mannose-binding protein deficiency - 614372
- ^ Toralf, Bernig; Willemijn, Breunis; Nannette, Brouwer; Amy, Hutchinson; Robert, Welch; Dirk, Roos; Taco, Kuijpers; Stephen, Chanock. "An analysis of genetic variation across the MBL2 locus in Dutch Caucasians indicates that 3' haplotypes could modify circulating levels of mannose-binding lectin". Hum Genet. 118 (3–4): 404. doi:10.1007/s00439-005-0053-5. PMID 16208516.
- ^ a b Colley KJ, Baenziger JU (1987). "Identification of the posttranslational modifications of the core-specific lectin. The core-specific lectin contains hydroxyproline, hydroxylysine, and glucosylgalactosylhydroxylysine residues". J Biol Chem. 262 (21): 10290–5. doi:10.1016/S0021-9258(18)61111-1. PMID 3611062.
- ^ "Mannose-binding protein C precursor [Homo sapiens]". Retrieved 2012-01-03.
- ^ Jensen PH, Laursen I, Matthiesen F, Højrup P (2007). "Posttranslational modifications in human plasma MBL and human recombinant MBL". Biochimica et Biophysica Acta (BBA) - Proteins and Proteomics. 1774 (3): 335–44. doi:10.1016/j.bbapap.2006.12.008. PMID 17289451.
- ^ Tomaiuolo R, Ruocco A, Salapete C, Carru C, Baggio G, Franceschi C, Zinellu A, Vaupel J, Bellia C, Lo Sasso B, Ciaccio M, Castaldo G, Deiana L (March 2012). "Activity of mannose-binding lectin (MBL) in centenarians". Aging Cell. 11 (3): 394–400. doi:10.1111/j.1474-9726.2012.00793.x. PMC 3935210. PMID 22239660.
- ^ Ogden CA, deCathelineau A, Hoffmann PR, Bratton D, Ghebrehiwet B, Fadok VA, Henson PM (September 2001). "C1q and mannose binding lectin engagement of cell surface calreticulin and CD91 initiates macropinocytosis and uptake of apoptotic cells". J. Exp. Med. 194 (6): 781–95. doi:10.1084/jem.194.6.781. PMC 2195958. PMID 11560994.
- ^ Stuart LM, Takahashi K, Shi L, Savill J, Ezekowitz RA (March 2005). "Mannose-binding lectin-deficient mice display defective apoptotic cell clearance but no autoimmune phenotype". J. Immunol. 174 (6): 3220–6. doi:10.4049/jimmunol.174.6.3220. PMID 15749852.
- ^ Choteau, L; Parny, M; François, N; Bertin, B; Fumery, M; Dubuquoy, L; Takahashi, K; Colombel, J-F; Jouault, T; Poulain, D; Sendid, B; Jawhara, S (7 October 2015). "Role of mannose-binding lectin in intestinal homeostasis and fungal elimination". Mucosal Immunology. 9 (3): 767–776. doi:10.1038/mi.2015.100. ISSN 1933-0219. PMID 26442658.
- ^ de Jong MA, Vriend LE, Theelen B, Taylor ME, Fluitsma D, Boekhout T, Geijtenbeek TB (March 2010). "C-type lectin Langerin is a beta-glucan receptor on human Langerhans cells that recognizes opportunistic and pathogenic fungi". Mol. Immunol. 47 (6): 1216–25. doi:10.1016/j.molimm.2009.12.016. PMC 2837148. PMID 20097424.
- ^ Ji X, Gewurz H, Spear GT (February 2005). "Mannose binding lectin (MBL) and HIV". Mol. Immunol. 42 (2): 145–52. doi:10.1016/j.molimm.2004.06.015. PMID 15488604.
- ^ Eriksson (2020). "Mannose-Binding Lectin is Associated with Thrombosis and Coagulopathy in Critically Ill COVID-19 Patients". Thrombosis and Haemostasis. 120 (12): 1720–1724. doi:10.1055/s-0040-1715835. PMC 7869044. PMID 32871607.
- ^ Stravalaci, Matteo; Pagani, Isabel; Paraboschi, Elvezia Maria; Pedotti, Mattia; Doni, Andrea; Scavello, Francesco; Mapelli, Sarah N.; Sironi, Marina; Perucchini, Chiara; Varani, Luca; Matkovic, Milos; Cavalli, Andrea; Cesana, Daniela; Gallina, Pierangela; Pedemonte, Nicoletta (February 2022). "Recognition and inhibition of SARS-CoV-2 by humoral innate immunity pattern recognition molecules". Nature Immunology. 23 (2): 275–286. doi:10.1038/s41590-021-01114-w. ISSN 1529-2908. PMID 35102342.
- ^ Herpers, B L; Endeman, H; de Jong, B A W; de Jongh, B M; Grutters, J C; Biesma, D H; vam Velzen-Blad, H (Jun 2009). "Acute-phase responsiveness of mannose-binding lectin in community-acquired pneumonia is highly dependent upon MBL2 genotypes". Clin Exp Immunol. 156 (3): 488–94. doi:10.1111/j.1365-2249.2009.03929.x. PMC 2691978. PMID 19438602.
- ^ Worthley DL, Bardy PG, Gordon DL, Mullighan CG (October 2006). "Mannose-binding lectin and maladies of the bowel and liver". World J. Gastroenterol. 12 (40): 6420–8. doi:10.3748/wjg.v12.i40.6420. PMC 4100630. PMID 17072973.
- ^ Hibberd, M. L.; Sumiya, M.; Summerfield, J. A.; Booy, R.; Levin, M. (1999). "Association of variants of the gene for mannose-binding lectin with susceptibility to meningococcal disease". The Lancet. 353 (9158): 1049–53. doi:10.1016/S0140-6736(98)08350-0. PMID 10199352. S2CID 6306870.
- ^ Faber, J.; Schuessler, T.; Finn, A.; Murdoch, C.; Zenz, W.; Habermehl, P.; Meyer, C. U.; Zabel, B. U.; Schmitt, H. J.; Zepp, F.; Knuf, M. (2007). "Age-Dependent Association of Human Mannose-Binding Lectin Mutations with Susceptibility to Invasive Meningococcal Disease in Childhood". The Pediatric Infectious Disease Journal. 26 (3): 243–246. doi:10.1097/01.inf.0000256751.76218.7c. PMID 17484222. S2CID 32019568.
- ^ Bradley, D. T.; Bourke, T. W.; Fairley, D. J.; Borrow, R.; Shields, M. D.; Young, I. S.; Zipfel, P. F.; Hughes, A. E. (2012). "Genetic susceptibility to invasive meningococcal disease: MBL2 structural polymorphisms revisited in a large case-control study and a systematic review". International Journal of Immunogenetics. 39 (4): 328–337. doi:10.1111/j.1744-313X.2012.01095.x. PMID 22296677. S2CID 205900750.[permanent dead link]
- ^ Thiel, S.; Frederiksen, P.D.; Jensenius, J.C. (January 2006). "Clinical manifestations of mannan-binding lectin deficiency". Molecular Immunology. 43 (1–2): 86–96. doi:10.1016/j.molimm.2005.06.018. PMC 7132399. PMID 16023210.