Polyaspartic acid (PASA) is a biodegradable, water-soluble condensation polymer based on the amino acid aspartic acid.[1][2] It is a biodegradable replacement for water softeners and related applications.[3] PASA can be chemically crosslinked with a wide variety of methods to yield PASA hydrogels.[4] The resulting hydrogels are pH-sensitive such that under acidic conditions, they shrink, while the swelling capacity increases under alkaline conditions.[4]
| |
| Names | |
|---|---|
| Other names
PASP
| |
| Identifiers | |
| |
| ChemSpider |
|
CompTox Dashboard (EPA)
|
|
| Properties | |
| (C4H5NO3)n | |
| Molar mass | variable |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |

Sodium polyaspartate is a sodium salt of polyaspartic acid.
In nature, PASA has been found in as fragments of larger proteins with length up to 50 amino acids,[5] but as of 2004 had not been isolated as a pure homo polymeric material from any natural source.[6] The first isolation of synthetic oligomeric sodium polyaspartate, obtained by thermal polycondensation of aspartic acid, was reported by Hugo Schiff in late 19th century.[7] Later it was proposed that thermal polymerization process leads through polysuccinimide intermediate.[8][9] Polyaspartic acid is produced industrially in both the acid form and as the sodium salt.[2]
Properties and structure
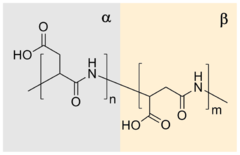
editDue to presence of carboxylic groups it is polyelectrolyte with anionic character. Naturally occurring PASA fragments consists of α,-linked L-aspartatic acid.[5] In contrast, the repeating unit of synthetic polyaspartic acid may exist in four isomeric forms, depending on the stereochemistry of starting material (D- and L-aspartic acid) and synthetic procedure leading to α and β links. Due to the protein-like backbone (presence of amide bond in the backbone), PASA has suitable biodegradability.[2]
Synthesis
editMany different routes lead to PASA. In the simplest[10] and the oldest approach[6] aspartic acid is heated to induce dehydration. In a subsequent step the resulting polysuccinimide is treated with aqueous sodium hydroxide, which yields partial opening of the succinimide rings. In this process sodium-DL-(α,β)-poly(aspartate) with 30% α-linkages and 70% β-linkages[11] randomly distributed along the polymer chain,[12] and racemized chiral center of aspartic acid is produced.[13] There were many catalysts reported for improving thermal polymerization method. Main benefits from their application is increasing of the conversion rate and higher molecular weight of the product.[14][15] Polyaspartic acid can also be synthesized by polymerization of maleic anhydride in presence of ammonium hydroxide.[1][2][16] High control over repeating unit isomers can be achieved by polymerization of N-carboxyanhydride (NCA) derivatives,[17] by polymerization of aspartic acid esters[18] or by application of enzyme catalyzed reaction.[19] Pure homopolymers, D- or L-PASA with α- or β-links only, can be synthesized using those methods.
The polymerization reaction is an example of a step-growth polymerization to a polyamide. In one procedure, aspartic acid polymerizes at 180 °C concomitant with dehydration and the formation of a poly(succinimide). The resulting polymer reacts with aqueous sodium hydroxide, which hydrolyzes one of the two amide bonds of the succinimide ring to form a sodium carboxylate. The remaining amide bond is thus the linkage between successive aspartate residues. Each aspartate residue is identified as α or β according to which carbonyl of it is part of the polymer chain. The α form has one carbon in the backbone in addition to the carbonyl itself (and a two-carbon sidechain) whereas the β form has two carbons in the backbone in addition to the carbonyl itself (and a one-carbon sidechain). This reaction gives a sodium poly(aspartate) composed of approximately 30% α-linkages and 70% β-linkages.[2]
Applications
editPolyaspartic acid and its derivatives are biodegradable alternatives to traditional polyanionic materials, in particular polyacrylic acid.[20] PASA has ability to inhibit deposition of calcium carbonate, calcium sulfate, barium sulfate, and calcium phosphate and can be used as an antiscaling agent in cooling water systems, water desalination processes, and waste water treatment operations.[21] In addition and due to its ability to chelate metal ions, it provides corrosion inhibition.[11] It can also be used as biodegradable detergent and dispersant for various applications.[22]
PASA also has a variety of biomedical applications. Its high affinity with calcium has been exploited for targeting various forms of drug-containing carriers to the bone.[2] The main component of bone is hydroxyapatite (ca. 70%) (mineralized calcium phosphate). Apart from bone targeting, PASA has been modified for other biomedical applications such as drug delivery, surface coating, DNA delivery, mucoadhesion, and beyond.[2]
As it can be synthesized in an environmentally friendly way and is biodegradable, polyaspartate is a potential green alternative to several materials such as sodium polyacrylate used in disposable diapers and agriculture.[23][24][25] It can act as a super-swelling material in diapers, feminine hygiene products, and food packaging.[26] The level of water uptake which is inversely related to the mechanical properties of the hydrogel can be tuned by changing the crosslinking density.[4]
See also
editReferences
edit- ^ a b Thomas Klein; Ralf-Johann Moritz; René Graupner (2008). "Polyaspartates and Polysuccinimide". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.l21_l01. ISBN 978-3527306732.
- ^ a b c d e f g Adelnia, Hossein; Tran, Huong D.N.; Little, Peter J.; Blakey, Idriss; Ta, Hang T. (2021-06-14). "Poly(aspartic acid) in Biomedical Applications: From Polymerization, Modification, Properties, Degradation, and Biocompatibility to Applications". ACS Biomaterials Science & Engineering. 7 (6): 2083–2105. doi:10.1021/acsbiomaterials.1c00150. hdl:10072/404497. PMID 33797239. S2CID 232761877.
- ^ Schwamborn, Michael (1998). "Chemical synthesis of polyaspartates: A biodegradable alternative to currently used polycar☐ylate homo- and copolymers". Polymer Degradation and Stability. 59 (1–3): 39–45. doi:10.1016/S0141-3910(97)00184-5.
- ^ a b c Adelnia, Hossein (2019). "Hydrogels Based on Poly (aspartic acid): Synthesis and Applications". Frontiers in Chemistry. 7: 755. Bibcode:2019FrCh....7..755A. doi:10.3389/fchem.2019.00755. PMC 6861526. PMID 31799235.
- ^ a b Rusenko, Kirt W.; Donachy, Julie E.; Wheeler, A. P. (1991). "Purification And Characterization Of A Shell Matrix Phosphoprotein From The American Oyster". In Sikes, C. Steven; Wheeler, A. P. (eds.). Surface Reactive Peptides and Polymers. ACS Symposium Series. Vol. 444. ACS. pp. 107–124. doi:10.1021/bk-1991-0444.ch008. ISBN 978-0-8412-1886-4.
- ^ a b Joentgen, Winfried; Müller, Nikolaus; Mitschker, Alfred; Schmidt, Holger (2004). "Polyaspartic acids". In Fahnestock, Stephen; Steinbüchel, Alexander (eds.). Polyamides and Complex Proteinaceous Materials I. Biopolymers. Vol. 7. Wiley-VCH. pp. 175–179. ISBN 978-3-527-30222-2.
- ^ Schiff, Hugo (1897). "Ueber Polyaspartsäuren". Ber. Dtsch. Chem. Ges. (in German). 30 (3): 2449–2459. doi:10.1002/cber.18970300316.
- ^ Kovács, J.; Könyves, I.; Pusztai, Á. (1953). "Darstellung von Polyasparaginsäuren (Polyaspartsäuren) aus dem thermischen Autokondensationsprodukt der Asparaginsäure". Experientia (in German). 9 (12): 459–460. doi:10.1007/BF02165821. PMID 13127859. S2CID 40153017.
- ^ Kovács, J.; Könyves, I. (1954). "Uber DL-α,β-Polyasparaginsaure". Naturwissenschaften (in German). 41 (14): 333. Bibcode:1954NW.....41..333K. doi:10.1007/BF00644501. S2CID 33648417.
- ^ Bennett, G. D. (2005). "A Green Polymerization of Aspartic Acid for the Undergraduate Organic Laboratory". Journal of Chemical Education. 82 (9): 1380–1381. Bibcode:2005JChEd..82.1380B. doi:10.1021/ed082p1380.
- ^ a b Low, Kim C.; Wheeler, A. P.; Koskan, Larry P. (1996). "6: Commercial Poly(aspartic acid) and Its Uses". In Glass, J. Edward (ed.). Hydrophilic Polymers. Advances in Chemistry. Vol. 248. ACS. pp. 99–111. doi:10.1021/ba-1996-0248.ch006. ISBN 978-0-8412-3133-7.
- ^ Pivcova, H.; Saudek, V.; Drobnik, J.; Vlasak, J. (1981). "NMR Study of Poly(aspartic acid). I. α-and β-Peptide Bonds in Poly(aspartic acid) Prepared by Thermal Polycondensation". Biopolymers. 20 (8): 1605–1614. doi:10.1002/bip.1981.360200804. S2CID 85201969.
- ^ Kokufuta, Etso; Suzuki, Shinnichiro; Harad, Kaoru (1978). "Temperature Effect on the Molecular Weight and the Optical Purity of Anhydropolyaspartic Acid Prepared by Thermal Polycondensation". Bulletin of the Chemical Society of Japan. 51 (5): 1555–1556. doi:10.1246/bcsj.51.1555.
- ^ Nakato, Takeshi; Kusuno, Atsushi; Kakuchi, Toyoji (2000). "Synthesis of poly(succinimide) by bulk polycondensation of L-aspartic acid with an acid catalyst". Journal of Polymer Science Part A: Polymer Chemistry. 38 (1): 117–122. Bibcode:2000JPoSA..38..117N. doi:10.1002/(SICI)1099-0518(20000101)38:1<117::AID-POLA15>3.0.CO;2-F.
- ^ Wang, Yaquan; Hou, Yongjiang; Ruan, Gang; Pan, Ming; Liu, Tengfei (2003). "Study on the polymerization of aspartic acid catalyzed by phosphoric acid". Journal of Macromolecular Science-Pure and Applied Chemistry. A40 (3): 293–307. doi:10.1081/MA-120018116. S2CID 85163135.
- ^ US patent 5468838, Boehmke, Gunter & Schmitz, Gerd, "Polysuccinimide, polyaspartic acid and their salts are prepared by reaction of maleic anhydride and ammonia, polycondensation of the resulting product in the presence of a solubilizing agent and, if appropriate, hydrolysis.", published 1995-11-21, assigned to Bayer AG
- ^ Rao, Vanga S.; Lapointe, Philippe; McGregor, Donald N. (1993). "Temperature Effect on the Molecular Weight and the Optical Purity of Anhydropolyaspartic Acid Prepared by Thermal Polycondensation". Makromolekulare Chemie-Macromolecular Chemistry and Physics. 194 (4): 1095–1104. doi:10.1002/macp.1993.021940405.
- ^ Saudek, V.; Pivcova, H.; Drobnik, J. (1981). "NMR Study of Poly(aspartic acid). II. α-and β-Peptide Bonds in Poly(aspartic acid) Prepared by Common Methods". Biopolymers. 20 (8): 1615–1623. doi:10.1002/bip.1981.360200805. S2CID 84203387.
- ^ Soeda, Yasuyuki; Toshima, Kazunobu; Matsuma, Shuichi (2003). "Sustainable enzymatic preparation of polyaspartate using a bacterial protease". Biomacromolecules. 4 (2): 193–203. doi:10.1021/bm0200534. PMID 12625712.
- ^ Gross, Richard A.; Kalra, Bhanu (2002). "Biodegradable Polymers for the Environment". Science. 297 (5582): 803–807. Bibcode:2002Sci...297..803G. doi:10.1126/science.297.5582.803. PMID 12161646.
- ^ Hasson, David; Shemer, Hilla; Sher, Alexander (2011). "State of the Art of Friendly "Green" Scale Control Inhibitors: A Review Article". Industrial & Engineering Chemistry Research. 50 (12): 7601–7607. doi:10.1021/ie200370v.
- ^ Thombre, Sunita M.; Sarwade, Bhimaro D. (2005). "Synthesis and Biodegradability of Polyaspartic Acid: A Critical Review" (PDF). Journal of Macromolecular Science, Part A. 42 (9): 1299–1315. doi:10.1080/10601320500189604. S2CID 94818855.
- ^ Gross, R. A.; Kalra, B. (2002). "Biodegradable Polymers for the Environment". Science. 297 (5582): 803–807. Bibcode:2002Sci...297..803G. doi:10.1126/science.297.5582.803. PMID 12161646.
- ^ "Presidential Green Chemistry Challenge Awards: 1996 Small Business Award: Donlar Corporation (now NanoChem Solutions, Inc.): Production and Use of Thermal Polyaspartic Acid". US Environmental Protection Agency.
- ^ Adelnia, Hossein; Blakey, Idriss; Little, Peter J.; Ta, Hang T. (2019). "Hydrogels Based on Poly(aspartic acid): Synthesis and Applications". Frontiers in Chemistry. 7: 755. Bibcode:2019FrCh....7..755A. doi:10.3389/fchem.2019.00755. ISSN 2296-2646. PMC 6861526. PMID 31799235.
- ^ Zahuriaan-Mehr, M. J.; Pourjavadi, A.; Salimi, H.; Kurdtabar, M. (2009). "Protein- and homo poly(amino acid)-based hydrogels with super-swelling properties". Polymers for Advanced Technologies. 20 (8): 655–671. doi:10.1002/pat.1395.