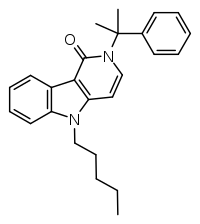
CUMYL-PEGACLONE (SGT-151) is a gamma-carboline based synthetic cannabinoid that has been sold as a designer drug.[3][4][5][6][7][8] The gamma-carboline core structure seen in CUMYL-PEGACLONE had not previously been encountered in a designer cannabinoid, though it is similar in structure to other gamma-carboline cannabinoids disclosed by Bristol-Myers Squibb in 2001.[9][10][11]
 | |
| Legal status | |
|---|---|
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| Chemical and physical data | |
| Formula | C25H28N2O |
| Molar mass | 372.512 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Legal status
editSweden's public health agency classified CUMYL-PEGACLONE as a narcotic substance, on January 18, 2019.[12]
In the United States, the DEA has temporarily placed CUMYL-PEGACLONE into Schedule I status starting on December 12th, 2023 for up to 2 years, during which it's possible the DEA could file for permanent scheduling within those 2 years. If the DEA does not file for permanent placement the temporary Schedule I order will expire on December 12th, 2025.[13]
See also
editReferences
edit- ^ Anvisa (2023-07-24). "RDC Nº 804 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 804 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 2023-07-25). Archived from the original on 2023-08-27. Retrieved 2023-08-27.
- ^ "Substance Details CUMYL-PEGACLONE". Retrieved 2024-01-22.
- ^ Ernst L, Brandhorst K, Papke U, Altrogge A, Zodel S, Langer N, Beuerle T (August 2017). "Identification and quantification of synthetic cannabinoids in 'spice-like' herbal mixtures: Update of the German situation in early 2017". Forensic Science International. 277: 51–58. doi:10.1016/j.forsciint.2017.05.019. PMID 28601726.
- ^ Angerer V, Mogler L, Steitz JP, Bisel P, Hess C, Schoeder CT, Müller CE, Huppertz LM, Westphal F, Schäper J, Auwärter V (July 2017). "Structural characterization and pharmacological evaluation of the new synthetic cannabinoid CUMYL-PEGACLONE". Drug Testing and Analysis. 10 (3): 597–603. doi:10.1002/dta.2237. PMID 28670781.
- ^ Mogler L, Wilde M, Huppertz LM, Weinfurtner G, Franz F, Auwärter V (January 2018). "Phase I metabolism of the recently emerged synthetic cannabinoid CUMYL-PEGACLONE and detection in human urine samples". Drug Testing and Analysis. 10 (5): 886–891. doi:10.1002/dta.2352. PMID 29314750.
- ^ Halter S, Angerer V, Röhrich J, Groth O, Roider G, Hermanns-Clausen M, Auwärter V (February 2019). "Cumyl-PEGACLONE: A comparatively safe new synthetic cannabinoid receptor agonist entering the NPS market?". Drug Testing and Analysis. 11 (2): 347–349. doi:10.1002/dta.2545. PMID 30468574. S2CID 53723006.
- ^ Janssens L, Cannaert A, Connolly MJ, Liu H, Stove CP (September 2020). "In vitro activity profiling of Cumyl-PEGACLONE variants at the CB1 receptor: Fluorination versus isomer exploration". Drug Testing and Analysis. 12 (9): 1336–1343. doi:10.1002/dta.2870. hdl:1854/LU-8687072. PMID 32490586. S2CID 219285656.
- ^ Tiemensma M, Rutherford JD, Scott T, Karch S (November 2020). "Emergence of Cumyl-PEGACLONE-related fatalities in the Northern Territory of Australia". Forensic Science, Medicine, and Pathology. 17 (1): 3–9. doi:10.1007/s12024-020-00334-0. PMID 33185835. S2CID 226309264.
- ^ WO application 2001058869, Leftheris K, Zhao, R, Chen BC, Kiener P, Wu H, Pandit C, Chennagiri R, Wrobleski S, Chen P, Hynes j, Longphre M, Norris D, Spergel S, Tokarski J, "Cannabinoid Receptor Modulators, Their Processes of Preparation, and Use of Cannabinoid Receptor Modulators in Treating Respiratory and Non-Respiratory Diseases", published 16 August 2001, assigned to Bristol-Myers Squibb Company
- ^ Wrobleski ST, Chen P, Hynes J, Lin S, Norris DJ, Pandit CR, Spergel S, Wu H, Tokarski JS, Chen X, Gillooly KM, Kiener PA, McIntyre KW, Patil-Koota V, Shuster DJ, Turk LA, Yang G, Leftheris K (May 2003). "Rational design and synthesis of an orally active indolopyridone as a novel conformationally constrained cannabinoid ligand possessing antiinflammatory properties". Journal of Medicinal Chemistry. 46 (11): 2110–6. doi:10.1021/jm020329q. PMID 12747783.
- ^ Alam RM, Keating JJ (March 2020). "Adding more "spice" to the pot: A review of the chemistry and pharmacology of newly emerging heterocyclic synthetic cannabinoid receptor agonists". Drug Testing and Analysis. 12 (3): 297–315. doi:10.1002/dta.2752. PMID 31854124. S2CID 209417068.
- ^ "Sexton nya ämnen klassas som narkotika eller hälsofarlig vara" (in Swedish). Folkhälsomyndigheten. 18 January 2019. Archived from the original on 3 June 2021. Retrieved 11 November 2019.
- ^ "Federal Register :: Request Access".