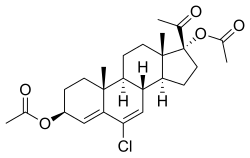
Clogestone acetate (USAN) (developmental code name AY-11440), also known as chlormadinol acetate or as 3β,17α-diacetoxy-6-chloropregna-4,6-diene-20-one,[1] is a steroidal progestin which was investigated as a progestin-only contraceptive and postcoital contraceptive but was never marketed.[2][3][4][5] It is the diacetate ester of clogestone, which, similarly was never marketed.[2] Clogestone acetate produces chlormadinone acetate as an active metabolite.[6]
 | |
| Clinical data | |
|---|---|
| Other names | Chlormadinol acetate; AY-11440; 3β,17α-Diacetoxy-6-chloropregna-4,6-diene-20-one |
| Drug class | Progestogen; Progestogen ester |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C25H33ClO5 |
| Molar mass | 448.98 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
See also
editReferences
edit- ^ Litwack G (2 December 2012). Biochemical Actions of Hormones. Elsevier. pp. 323–. ISBN 978-0-323-15189-4.
- ^ a b Elks J (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 297–. ISBN 978-1-4757-2085-3.
- ^ Hawkins DR, Elder MG (22 October 2013). Human Fertility Control: Theory and Practice. Elsevier Science. pp. 3–. ISBN 978-1-4831-6361-1.
- ^ Harper MJ (8 March 2013). "Contraception — retrospect and prospect". Progress in Drug Research, vol. 21. Progress in Drug Research / Fortschritte der Arzneimittelforschung / Progrès des rechersches pharmaceutiques. Vol. 21. Birkhäuser. pp. 293–407. doi:10.1007/978-3-0348-7098-6_4. ISBN 978-3-0348-7098-6. PMID 339271.
- ^ Hinselmann M, Jürgensen O, Hasselblatt I, Otten U, Prinz W, Taubert HD (August 1970). "[Clogestone acetate, a new orally effective progestagen]". Archiv Fur Gynakologie (in German). 209 (2): 136–148. doi:10.1007/BF00668180. PMID 5537778.
- ^ Stern MD, Givner ML (April 1975). "Measurement of serum clogestone acetate (AY-11,440) by a radioreceptor assay: a practical approach to the quantitative determination of synthetic progestins". The Journal of Clinical Endocrinology and Metabolism. 40 (4): 728–731. doi:10.1210/jcem-40-4-728. PMID 1127082.