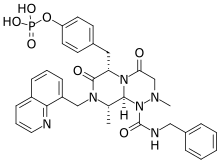
Foscenvivint (PRI 724 or OP 724)[1] is a CREB-binding protein/β-catenin inhibitor that is developed for the treatment of liver diseases[2] such as primary biliary cholangitis,[3] hepatocellular carcinoma,[4][5] and hepatitis C and B virus-induced liver cirrhosis.[6][7]
 | |
| Clinical data | |
|---|---|
| Other names | PRI 724; OP 724 |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| UNII | |
| Chemical and physical data | |
| Formula | C33H35N6O7P |
| Molar mass | 658.652 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
References
edit- ^ "Foscenvivint - PRISM Pharma Co - AdisInsight". adisinsight.springer.com. Retrieved 23 November 2023.
- ^ Ouchi, Hirofumi; Mizutani, Yuki; Yoshimura, Kaori; Sato, Yasunori; Kimura, Kiminori; Haruyama, Yushi; Harada, Kenichi (June 2023). "Anti-inflammatory and antifibrotic effects of CBP/β-catenin inhibitor for hepatocytes: small molecular inhibitor, OP-724 possibly improves liver function". Medical Molecular Morphology. 56 (2): 94–105. doi:10.1007/s00795-022-00343-8. PMID 36645521. S2CID 255847780.
- ^ Kimura, Masamichi; Ogawa, Eiichi; Harada, Kenichi; Imamura, Jun; Saio, Masanao; Ikura, Yoshihiro; Yatsuhashi, Hiroshi; Murata, Kazumoto; Miura, Kouichi; Ieiri, Ichiro; Tanaka, Atsushi; Kimura, Kiminori (1 November 2022). "Feasibility, safety and tolerability of the CREB-binding protein/β-catenin inhibitor OP-724 in patients with advanced primary biliary cholangitis: an investigator-initiated, open-label, non-randomised, two-centre, phase 1 study". BMJ Open Gastroenterology. 9 (1): e001001. doi:10.1136/bmjgast-2022-001001. ISSN 2054-4774. PMC 9710334. PMID 36442892.
- ^ El-Khoueiry, Anthony B.; Ning, Yan; Yang, Dongyun; Cole, Sarah; Kahn, Michael; Zoghbi, Marwan; Berg, Jennifer; Fujimori, Masamoto; Inada, Tetsuhi; Kouji, Hiroyuki; Lenz, Heinz-Josef (20 May 2013). "A phase I first-in-human study of PRI-724 in patients (pts) with advanced solid tumors". Journal of Clinical Oncology. 31 (15_suppl): 2501. doi:10.1200/jco.2013.31.15_suppl.2501.
- ^ Gabata, Ryosuke; Harada, Kenichi; Mizutani, Yuki; Ouchi, Hirofumi; Yoshimura, Kaori; Sato, Yasunori; Kitao, Azusa; Kimura, Kiminori; Kouji, Hiroyuki; Miyashita, Tomoharu; Tajima, Hidehiro; Ohta, Tetsuo (September 2020). "Anti-tumor Activity of the Small Molecule Inhibitor PRI-724 Against β-Catenin-activated Hepatocellular Carcinoma". Anticancer Research. 40 (9): 5211–5219. doi:10.21873/anticanres.14524. PMID 32878809.
- ^ Kimura, Kiminori; Kanto, Tatsuya; Shimoda, Shinji; Harada, Kenichi; Kimura, Masamichi; Nishikawa, Koji; Imamura, Jun; Ogawa, Eiichi; Saio, Masanao; Ikura, Yoshihiro; Okusaka, Takuji; Inoue, Kazuaki; Ishikawa, Tetsuya; Ieiri, Ichiro; Kishimoto, Junji; Todaka, Koji; Kamisawa, Terumi (June 2022). "Safety, tolerability, and anti-fibrotic efficacy of the CBP/β-catenin inhibitor PRI-724 in patients with hepatitis C and B virus-induced liver cirrhosis: An investigator-initiated, open-label, non-randomised, multicentre, phase 1/2a study". eBioMedicine. 80: 104069. doi:10.1016/j.ebiom.2022.104069. PMC 9126795. PMID 35605429.
- ^ Kimura, Kiminori; Ikoma, Akemi; Shibakawa, Maki; Shimoda, Shinji; Harada, Kenichi; Saio, Masanao; Imamura, Jun; Osawa, Yosuke; Kimura, Masamichi; Nishikawa, Koji; Okusaka, Takuji; Morita, Satoshi; Inoue, Kazuaki; Kanto, Tatsuya; Todaka, Koji; Nakanishi, Yoichi; Kohara, Michinori; Mizokami, Masashi (September 2017). "Safety, Tolerability, and Preliminary Efficacy of the Anti-Fibrotic Small Molecule PRI-724, a CBP/β-Catenin Inhibitor, in Patients with Hepatitis C Virus-related Cirrhosis: A Single-Center, Open-Label, Dose Escalation Phase 1 Trial". eBioMedicine. 23: 79–87. doi:10.1016/j.ebiom.2017.08.016. PMC 5605374. PMID 28844410.