Lenacapavir, sold under the brand name Sunlenca, is an antiretroviral medication used to treat HIV/AIDS.[9][10] It is taken by mouth or by subcutaneous injection.[9][10]
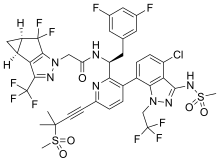 | |
| Clinical data | |
|---|---|
| Trade names | Sunlenca |
| Other names | GS-CA1, GS-6207 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth, subcutaneous |
| Drug class | Capsid inhibitors |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| |
| CAS Number | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| PDB ligand | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C39H32ClF10N7O5S2 |
| Molar mass | 968.28 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
The most common side effects include reactions at the injection site and nausea.[10][11]
Lenacapavir was approved for medical treatment in the European Union in August 2022,[10][12] in Canada in November 2022,[5][6] and in the United States in December 2022.[9][11][13][14] It is the first of a class of drugs called capsid inhibitors to be approved by the US Food and Drug Administration (FDA) for treating HIV/AIDS.[11][15]
Medical uses
editLenacapavir, in combination with other antiretrovirals, is indicated for the treatment of HIV/AIDS.[9] It is used in heavily treatment-experienced adults with multiple drug resistance in whom current antiretroviral therapy is ineffective due to resistance, intolerance or safety considerations.[9][11] It has also been found to be highly effective as HIV pre-exposure prophylaxis (PrEP) in heterosexual women in Africa.[16] Further studies are ongoing assessing effectiveness in men who have sex with men and people who inject drugs.[17]
Mechanism of action
editLenacapavir works by binding directly to the interface between HIV-1 viral capsid protein (p24) subunits in capsid hexamers, interfering with essential steps of viral replication, including capsid-mediated nuclear uptake of HIV-1 proviral DNA[clarification needed], virus assembly and release, production of capsid protein subunits, and capsid core formation.[9][11] The US Food and Drug Administration considers it to be a first-in-class medication.[15][18]
History
editLenacapavir was developed by Gilead Sciences.[19]
The safety and efficacy of lenacapavir were established through a multicenter clinical trial with 72 participants whose HIV infections were resistant to multiple classes of HIV medications.[11] These participants had to have high levels of virus in their blood despite being on antiretroviral drugs.[11] Participants were enrolled into one of two study groups.[11] One group was randomized to receive either lenacapavir or placebo in a double-blind fashion, and the other group received open-label lenacapavir.[11] The primary measure of efficacy was the proportion of participants in the randomized study group who achieved a certain level of reduction in virus during the initial 14 days compared to baseline.[11]
The US Food and Drug Administration granted the application for lenacapavir priority review, fast track, and breakthrough therapy designations.[11]
Society and culture
editLegal status
editIn June 2022, the Committee for Medicinal Products for Human Use of the European Medicines Agency adopted a positive opinion, recommending the granting of a marketing authorization for the medicinal product Sunlenca, intended for the treatment of adults with multidrug‑resistant human immunodeficiency virus type 1 (HIV‑1) infection.[20] The applicant for this medicinal product is Gilead Sciences Ireland UC.[20]
Lenacapavir was approved for medical use in the European Union in August 2022,[10][21] in Canada in November 2022,[5][6] and in the United States in December 2022.[11]
Economics
editAs of 2024[update] the drug, produced by Gilead, costs US$42,250 for the first year. A study presented in July 2024[22] found that mass production of a generic version would allow a profit margin of 30% on an annual price of $40 if used by 10 million people. The authors said that lowering HIV levels significantly would probably require 60 million people to take the drug preventatively.[23]
Research
editAs of 2021, it is in phase II/III clinical trials.[24] It is being investigated as a treatment for HIV patients infected with multidrug-resistant virus and as a twice-yearly injectable for pre-exposure prophylaxis.[24][25]
Studies have been conducted for the use of lenacapavir in treatment-naive individuals.[26] For virally suppressed individuals switching treatment, early studies have tested lenacapavir injections in combination with infusions of the broadly neutralizing antibodies teropavimab and zinlirvimab[27] as well as lenacapavir with islatravir.[28]
In July 2024, a study reported 100% efficacy for pre-exposure HIV prevention (PrEP). The randomized phase 3 clinical trial involved 5,338 young women in South Africa and Uganda. All 2,134 women who received an injection every six months remained HIV-free. In contrast, groups taking other daily oral PrEP drugs still had HIV cases, likely due to low adherence.[29][30][31][32]
References
edit- ^ a b "Sunlenca". Therapeutic Goods Administration. 6 April 2023. Archived from the original on 8 April 2023. Retrieved 7 April 2023.
- ^ "Sunlenca lenacapavir (as sodium) 300 mg film coated tablet blister pack (392350)". Therapeutic Goods Administration. 28 March 2023. Archived from the original on 8 April 2023. Retrieved 7 April 2023.
- ^ "Sunlenca lenacapavir (as sodium) 463.5 mg/1.5 mL solution for injection vial (386895)". Therapeutic Goods Administration. 28 March 2023. Archived from the original on 8 April 2023. Retrieved 7 April 2023.
- ^ https://www.tga.gov.au/resources/auspar/auspar-sunlenca [bare URL]
- ^ a b c "Sunlenca Oral Product information". Health Canada. 25 April 2012. Archived from the original on 15 January 2023. Retrieved 23 December 2022.
- ^ a b c "Sunlenca Subcutaneous Product information". Health Canada. 25 April 2012. Archived from the original on 15 January 2023. Retrieved 23 December 2022.
- ^ "Summary Basis of Decision - Sunlenca". Health Canada. 10 March 2023. Archived from the original on 25 April 2023. Retrieved 24 April 2023.
- ^ "Details for: Sunlenca". Health Canada. 15 March 2023. Archived from the original on 3 March 2024. Retrieved 3 March 2024.
- ^ a b c d e f g "Sunlenca- lenacapavir sodium tablet, film coated Sunlenca- lenacapavir sodium kit". DailyMed. 21 December 2022. Archived from the original on 21 January 2023. Retrieved 21 January 2023.
- ^ a b c d e f "Sunlenca EPAR". European Medicines Agency (EMA). 22 June 2022. Archived from the original on 26 August 2022. Retrieved 25 August 2022. Text was copied from this source which is copyright European Medicines Agency. Reproduction is authorized provided the source is acknowledged.
- ^ a b c d e f g h i j k l "FDA Approves New HIV Drug for Adults with Limited Treatment Options" (Press release). U.S. Food and Drug Administration (FDA). 22 December 2022. Archived from the original on 15 January 2023. Retrieved 23 December 2022. This article incorporates text from this source, which is in the public domain.
- ^ "Gilead Announces First Global Regulatory Approval of Sunlenca (Lenacapavir), the Only Twice-Yearly HIV Treatment Option". Gilead Sciences, Inc. (Press release). 22 August 2022. Archived from the original on 15 January 2023. Retrieved 23 December 2022.
- ^ "Sunlenca (lenacapavir) Receives FDA Approval as a First-in-Class, Twice-Yearly Treatment Option for People Living With Multi-Drug Resistant HIV". Gilead Sciences, Inc. (Press release). 22 December 2022. Archived from the original on 23 December 2022. Retrieved 23 December 2022.
- ^ Paik J (September 2022). "Lenacapavir: First Approval". Drugs. 82 (14): 1499–1504. doi:10.1007/s40265-022-01786-0. PMC 10267266. PMID 36272024.
- ^ a b "Advancing Health Through Innovation: New Drug Therapy Approvals 2022". U.S. Food and Drug Administration. 10 January 2023. Archived from the original on 21 January 2023. Retrieved 22 January 2023. This article incorporates text from this source, which is in the public domain.
- ^ Bekker LG, Das M, Abdool Karim Q, Ahmed K, Batting J, Brumskine W, et al. (July 2024). "Twice-Yearly Lenacapavir or Daily F/TAF for HIV Prevention in Cisgender Women". The New England Journal of Medicine. 391 (13): 1179–1192. doi:10.1056/NEJMoa2407001. PMID 39046157.
- ^ "Prevention With a Purpose". purposestudies.com.
- ^ New Drug Therapy Approvals 2022 (PDF). U.S. Food and Drug Administration (FDA) (Report). January 2024. Archived from the original on 14 January 2024. Retrieved 14 January 2024. This article incorporates text from this source, which is in the public domain.
- ^ Link JO, Rhee MS, Tse WC, Zheng J, Somoza JR, Rowe W, et al. (August 2020). "Clinical targeting of HIV capsid protein with a long-acting small molecule". Nature. 584 (7822): 614–618. Bibcode:2020Natur.584..614L. doi:10.1038/s41586-020-2443-1. PMC 8188729. PMID 32612233. S2CID 220293679.
- ^ a b "Sunlenca: Pending EC decision". European Medicines Agency. 23 June 2022. Archived from the original on 26 June 2022. Retrieved 26 June 2022. Text was copied from this source which is copyright European Medicines Agency. Reproduction is authorized provided the source is acknowledged.
- ^ "Sunlenca Product information". Union Register of medicinal products. Archived from the original on 3 March 2023. Retrieved 3 March 2023.
- ^ "AIDS 2024, the 25th International AIDS Conference". International AIDS Society (IAS). Retrieved 23 July 2024.
- ^ Lay K (23 July 2024). "HIV 'vaccine' could be made for just $40 a year for every patient". The Guardian.
- ^ a b Boerner H (11 March 2021). "Lenacapavir Effective in Multidrug Resistant HIV". Medscape. Archived from the original on 16 March 2021. Retrieved 15 March 2021.
- ^ Highleyman L (15 March 2021). "Lenacapavir Shows Promise for Long-Acting HIV Treatment and Prevention". POZ. Archived from the original on 19 July 2021.
- ^ Gupta S (January 2023). "Lenacapavir administered every 26 weeks or daily in combination with oral daily antiretroviral therapy for initial treatment of HIV: a randomised, open-label, active-controlled, phase 2 trial". The Lancet HIV. 10 (1): 15–23. doi:10.1016/S2352-3018(22)00291-0. PMID 36566079. Retrieved 10 March 2024.
- ^ Highleyman L (28 February 2023). "Lenacapavir plus broadly neutralising antibodies may offer twice-yearly treatment option". Aidsmap. Retrieved 10 March 2024.
- ^ Highleyman L (6 March 2024). "Islatravir plus lenacapavir could be the first once-weekly oral HIV treatment". Aidsmap. Retrieved 10 March 2024.
- ^ Bekker LG, Das M, Abdool Karim Q, Ahmed K, Batting J, Brumskine W, et al. (July 2024). "Twice-Yearly Lenacapavir or Daily F/TAF for HIV Prevention in Cisgender Women". The New England Journal of Medicine. 391 (13): 1179–1192. doi:10.1056/NEJMoa2407001. PMID 39046157.
- ^ "Lenacapavir Shows 100% Efficacy and Zero Infections in HIV Prevention". www.precisionvaccinations.com. Retrieved 25 July 2024.
- ^ "Full Efficacy and Safety Results for Gilead Investigational Twice-Yearly Lenacapavir for HIV Prevention Presented at AIDS 2024". www.gilead.com. Retrieved 25 July 2024.
- ^ Bekker LG (3 July 2024). "HIV breakthrough: drug trial shows injection twice a year is 100% effective against infection". The Conversation. Retrieved 25 July 2024.
External links
edit- "Lenacapavir". Clinical Info. National Institutes of Health.