Pentyl is a five-carbon alkyl group or substituent with chemical formula -C5H11. It is the substituent form of the alkane pentane.
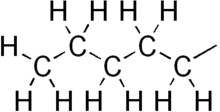
| |
| Identifiers | |
|---|---|
3D model (JSmol)
|
|
| ChemSpider | |
PubChem CID
|
|
| |
| |
| Properties | |
| −C5H11 | |
| Molar mass | 71.1408 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
In older literature, the common non-systematic name amyl was often used for the pentyl group. Conversely, the name pentyl was used for several five-carbon branched alkyl groups, distinguished by various prefixes. The nomenclature has now reversed, with "amyl" being more often used to refer to the terminally branched group also called isopentyl, as in amobarbital.
A cyclopentyl group is a ring with the formula -C5H9.
The name is also used for the pentyl radical, a pentyl group as an isolated molecule. This free radical is only observed in extreme conditions.[1] Its formula is often written "C
5H
11•" or "• C
5H
11" to indicate that it has one unsatisfied valence bond. Radicals like pentyl are reactive, they react with neighboring atoms or molecules (like oxygen, water, etc.)
Older "pentyl" groups
editThe following names are still used sometimes:
| Name | Structure | IUPAC status | IUPAC recomm. | Examples |
|---|---|---|---|---|
| n-pentyl, amyl | H 3C−(CH 2) 4− |
pentyl | [2] | |
| tert-pentyl | H 3C−CH 2−(H 3C−) 2C− |
No longer recommended[3] | 2-methylbutan-2-yl (aka 1,1-dimethylpropyl)[3] | [4] |
| neopentyl | (H 3C−) 3C−CH 2− |
No longer recommended[3] | 2,2-dimethylpropyl[3] | [5] |
| isopentyl, amyl, isoamyl | (H 3C−) 2CH−(CH 2) 2− |
No longer recommended[3] | 3-methylbutyl[3] | [6] |
| sec-pentyl | H 3C−(CH 2) 2−(H 3C−)CH− |
pentan-2-yl[7](or (1-methylbutyl)) | [8] | |
| 3-pentyl | (H 3C−CH 2−) 2CH− |
pentan-3-yl (also known as 1-ethylpropyl) | [9] | |
| sec-isopentyl | (H 3C−) 2CH−(H 3C−)CH− |
3-methylbutan-2-yl (or 1,2-dimethylpropyl) | ||
| active pentyl | H 3C−CH 2−(H 3C−)CH−CH 2− |
2-methylbutyl |
Pentyl radical
editThe free radical pentyl was studied by J. Pacansky and A. Gutierrez in 1983.[1] The radical was obtained by exposing bishexanoyl peroxide trapped in frozen argon to ultraviolet light, that caused its decomposition into two carbon dioxide (CO
2) molecules and two pentyl radicals.
Examples
editReferences
edit- ^ a b J. Pacansky , A. Gutierrez (1983), "Infrared spectra of the n-butyl and n-pentyl radicals". Journal of Physical Chemistry volume 87, issue 16, pages 3074–3079doi:10.1021/j100239a023
- ^ n-pentyl nitrite, described as "a nitrite ester having n-pentyl as the alkyl group." Accessed on 2013-02-21
- ^ a b c d e f Nomenclature of Organic Chemistry : IUPAC Recommendations and Preferred Names 2013 (Blue Book). Cambridge: The Royal Society of Chemistry. 2014. p. 607. doi:10.1039/9781849733069-FP001. ISBN 978-0-85404-182-4.
- ^ NCBI, tert-pentyl alcohol in PubChem. Accessed on 2013-02-21
- ^ neopentyl at CHEBI. Accessed on 2013-02-21
- ^ isopentyl group at CHEBI. Accessed on 2013-02-21
- ^ Nomenclature of Organic Chemistry : IUPAC Recommendations and Preferred Names 2013 (Blue Book). Cambridge: The Royal Society of Chemistry. 2014. p. 362. doi:10.1039/9781849733069-FP001. ISBN 978-0-85404-182-4.
- ^ Pentobarbital at CHEBI, described as "Barbituric acid substituted at C-5 by ethyl and sec-pentyl groups". Accessed on 2013-02-21
- ^ Pentan-3-yl group at CHEBI. Accessed on 2013-02-21