The inorganic dye ammoniated ruthenium oxychloride, also known as ruthenium red, is used in histology to stain aldehyde fixed mucopolysaccharides.
| |
| Identifiers | |
|---|---|
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.228.922 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
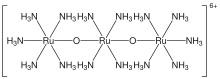
| Cl6H42N14O2Ru3 | |
| Molar mass | 786.34 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Ruthenium red (RR) has also been used as a pharmacological tool to study specific cellular mechanisms. Selectivity is a significant issue in such studies as RR is known to interact with many proteins.[1] These include mammalian ion channels (CatSper1, TASK, RyR1, RyR2, RyR3, TRPM6, TRPM8, TRPV1, TRPV2, TRPV3, TRPV4, TRPV5, TRPV6, TRPA1, mCa1, mCa2, CALHM1[2][3]) TRPP3,[4] a plant ion channel, Ca2+-ATPase, mitochondrial Ca2+ uniporter,[5] tubulin, myosin light-chain phosphatase, and Ca2+ binding proteins such as calmodulin. Ruthenium red displays nanomolar potency against several of its binding partners (e.g. TRPV4, ryanodine receptors,...). For example, it is a potent inhibitor of intracellular calcium release by ryanodine receptors (Kd ~20 nM).[6] As a TRPA1 blocker, it assists in reducing the airway inflammation caused by pepper spray.
RR has been used on plant material since 1890 for staining pectins, mucilages, and gums. RR is a stereoselective stain for pectic acid, insofar as the staining site occurs between each monomer unit and the next adjacent neighbor.[7]
References
edit- ^ Vincent, F; Duncton, MA (2011). "TRPV4 agonists and antagonists". Curr Top Med Chem. 11 (17): 2216–26. doi:10.2174/156802611796904861. PMID 21671873.
- ^ Ma, Z; Siebert, AP; Cheung, KH; Lee, RJ; Johnson, B; Cohen, AS; Vingtdeux, V; Marambaud, P; Foskett, JK (2012). "Calcium homeostasis modulator 1 (CALHM1) is the pore-forming subunit of an ion channel that mediates extracellular Ca2+ regulation of neuronal excitability". Proc Natl Acad Sci USA. 109 (28): E1963–71. Bibcode:2012PNAS..109E1963M. doi:10.1073/pnas.1204023109. PMC 3396471. PMID 22711817.
- ^ Dreses-Werringloer, U; Vingtdeux, V; Zhao, H; Chandakkar, P; Davies, P; Marambaud, P (2013). "CALHM1 controls Ca2+-dependent MEK/ERK/RSK/MSK signaling in neurons". J Cell Sci. 126 (Pt 5): 1199–206. doi:10.1242/jcs.117135. PMC 4481642. PMID 23345406.
- ^ Decaen, P. G.; Delling, M.; Vien, T. N.; Clapham, D. E. (2013). "Direct recording and molecular identification of the calcium channel of primary cilia". Nature. 504 (7479): 315–318. Bibcode:2013Natur.504..315D. doi:10.1038/nature12832. PMC 4073646. PMID 24336289.
- ^ Hajnóczky, G; Csordás, G; Das, S; Garcia-Perez, C; Saotome, M; Sinha Roy, S; Yi, M (2006). "Mitochondrial calcium signalling and cell death: approaches for assessing the role of mitochondrial Ca2+ uptake in apoptosis". Cell Calcium. 40 (5–6): 553–60. doi:10.1016/j.ceca.2006.08.016. PMC 2692319. PMID 17074387.
- ^ Tripathy, Le Xu Ashutosh; Pasek, Daniel A.; Meissner, Gerhard (1998). "Potential for Pharmacology of Ryanodine Receptor/Calcium Release Channels". Ann NY Acad Sci. 853 (1): 130–148. Bibcode:1998NYASA.853..130T. doi:10.1111/j.1749-6632.1998.tb08262.x. PMID 10603942. S2CID 86436194. Archived from the original on 2008-04-23. Retrieved 2006-10-22.
- ^ Mariani Colombo P, Rascio N. "Ruthenium red staining for electron microscopy of plant material". Journal of Ultrastructure Research Volume 60, Issue 2, August 1977, Pages 135–139