 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Enablex, Emselex, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a605039 |
| Pregnancy category |
|
| Routes of administration | By mouth |
| Drug class | Anticholinergic[1] |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 15 to 19% (dose-dependent) |
| Protein binding | 98% |
| Metabolism | Liver (CYP2D6- and CYP3A4-mediated) |
| Elimination half-life | 13 to 19 hours |
| Excretion | Kidney (60%) and biliary (40%) |
| Identifiers | |
| |
| Chemical and physical data | |
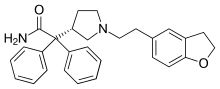
| Formula | C28H30N2O2 |
| Molar mass | 426.560 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Darifenacin, sold under the trade name Enablex among others, is a medication used to treat overactive bladder.[2] It is taken once per day by mouth.[2] Benefits are generally seen within 2 weeks.[2]
Common side effects include dry mouth and constipation.[2] Other side effects may include cough, bladder pain, blurry vision, urinary retention, shortness of breath, sexual dysfunction, and high blood pressure.[2][3] Use is not recommended during pregnancy.[3] It is an anticholinergic which helps the bladder relax.[1]
Darifenacin was approved for medical use in the United States and Europe in 2004.[2][1] It is available as a generic medication.[4] In the United States it costs about 37 USD per month as of 2021.[4] This amount in the United Kingdom costs the NHS about £25.[3]
References
edit- ^ a b c "Emselex". Archived from the original on 10 January 2021. Retrieved 21 July 2021.
- ^ a b c d e f g h "Darifenacin Monograph for Professionals". Drugs.com. Archived from the original on 18 January 2021. Retrieved 21 July 2021.
- ^ a b c BNF (80 ed.). BMJ Group and the Pharmaceutical Press. September 2020 – March 2021. p. 821. ISBN 978-0-85711-369-6.
{{cite book}}: CS1 maint: date format (link) - ^ a b "Darifenacin ER Prices, Coupons & Savings Tips - GoodRx". GoodRx. Archived from the original on 4 July 2016. Retrieved 21 July 2021.