 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Synribo, Tekinex, others |
| Other names | Omacetaxine mepesuccinate, homoharringtonine (HHT) |
| AHFS/Drugs.com | Monograph |
| License data | |
| Routes of administration | Subcutaneous, intravenous infusion |
| Drug class | Protein synthesis inhibitor[1] |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Protein binding | 50% |
| Metabolism | Mostly via plasma esterases |
| Elimination half-life | 6 hours |
| Excretion | Urine (≤15% unchanged) |
| Identifiers | |
| |
| Chemical and physical data | |
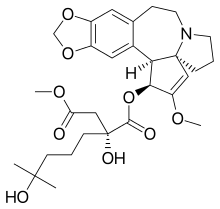
| Formula | C29H39NO9 |
| Molar mass | 545.629 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Omacetaxine, sold under the brand name Synribo, is a medication used to treat chronic myeloid leukemia (CML).[1] It is used in cases were other medications have not worked.[1] It is given by injection under the skin.[1]
Common side effects include low platelets, low red blood cells, low white blood cells, fever, diarrhea, nausea, and pain.[1] Other side effects may include bleeding and infection.[1] Use during pregnancy may harm the baby.[1] It is a protein synthesis inhibitor.[1]
Omacetaxine was approved for medical use in the United States in 2012.[1] It is not approved in Europe.[2] In the United States it costs about 1,200 USD for 3.5 mg as of 2021.[3] It is made from a substance in the Chinese evergreen known as harringtonine.[2]
References
edit- ^ a b c d e f g h i j k l "Omacetaxine Monograph for Professionals". Drugs.com. Archived from the original on 4 March 2021. Retrieved 7 November 2021.
- ^ a b "Tekinex". Archived from the original on 30 October 2020. Retrieved 7 November 2021.
- ^ "Synribo Prices, Coupons & Patient Assistance Programs". Drugs.com. Retrieved 7 November 2021.