XPhos is a phosphine ligand derived from biphenyl. Its palladium complexes exhibit high activity for Buchwald-Hartwig amination reactions involving aryl chlorides and aryl tosylates. Both palladium and copper complexes of the compound exhibit high activity for the coupling of aryl halides and aryl tosylates with various amides.[1] It is also an efficient ligand for several commonly used C–C bond-forming cross-coupling reactions, including the Negishi, Suzuki, and the copper-free Sonogashira coupling reactions. It is especially efficient and general when employed as a (2-aminobiphenyl)-cyclometalated palladium mesylate precatalyst complex (Buchwald's third generation precatalyst system), XPhos-G3-Pd, which is commercially available and stable to bench storage.[2][3][4] The ligand itself also has convenient handling characteristics as a crystalline, air-stable solid.[5]
| |
| Names | |
|---|---|
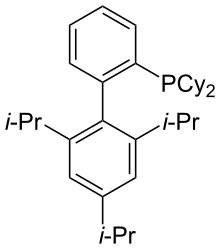
| Preferred IUPAC name
Dicyclohexyl[2′,4′,6′-tris(propan-2-yl)[1,1′-biphenyl]-2-yl]phosphane | |
| Other names
XPhos
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.123.428 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C33H49P | |
| Molar mass | 476.72 |
| Appearance | colorless solid |
| Melting point | 187 to 190 °C (369 to 374 °F; 460 to 463 K) |
| organic solvents | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Structure
edit-
One view of the molecule's structure.
-
Another view of the molecule's structure.
See also
editReferences
edit- ^ Huang, X.; Anderson, K. W.; Zim, D.; Jiang, L.; Klapars, A.; Buchwald, S. L. (2003). "Expanding Pd-Catalyzed C-N Bond-Forming Processes: The First Amidation of Aryl Sulfonates, Aqueous Amination, and Complementarity with Cu-Catalyzed Reactions". J. Am. Chem. Soc. 125 (22): 6653–6655. doi:10.1021/ja035483w. PMID 12769573.
- ^ Bruno, Nicholas C.; Tudge, Matthew T.; Buchwald, Stephen L. (2013-02-04). "Design and preparation of new palladium precatalysts for C–C and C–N cross-coupling reactions". Chemical Science. 4 (3): 916–920. doi:10.1039/C2SC20903A. ISSN 2041-6539. PMC 3647481. PMID 23667737.
- ^ Yang, Yang; Oldenhuis, Nathan J.; Buchwald, Stephen L. (2013). "Mild and General Conditions for Negishi Cross-Coupling Enabled by the Use of Palladacycle Precatalysts". Angew. Chem. Int. Ed. 52 (2): 615–619. doi:10.1002/anie.201207750. PMC 3697109. PMID 23172689.
- ^ Gelman, Dmitri; Buchwald, Stephen L. (2003). "Efficient Palladium-Catalyzed Coupling of Aryl Chlorides and Tosylates with Terminal Alkynes: Use of a Copper Cocatalyst Inhibits the Reaction". Angew. Chem. Int. Ed. 42 (48): 5993–5996. doi:10.1002/anie.200353015. PMID 14679552.
- ^ Altman, R.A.; Fors, B.P.; Buchwald, S.L. (2007). "Pd-Catalyzed Amination Reactions of Aryl Halides Using Bulky Biarylmonophosphine Ligands". Nature Protocols. 2 (11): 2881–2887. doi:10.1038/nprot.2007.414. PMID 18007623. S2CID 23134711.