Selegiline, also known as L-deprenyl and sold under the brand names Eldepryl, Zelapar, and Emsam among others, is a medication which is used in the treatment of Parkinson's disease and major depressive disorder.[1] It is provided in the form of a capsule or tablet taken by mouth or orally disintegrating tablets taken on the tongue for Parkinson's disease and as a patch applied to skin for depression.
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /səˈlɛdʒɪliːn/ sə-LEJ-i-leen |
| Trade names | Eldepryl, Jumex, Zelapar, Emsam, others[1] |
| Other names | L-Deprenyl; (R)-(–)-N,α-Dimethyl-N-2-propynylphenethylamine; (R)-(–)-N-Methyl-N-2-propynylamphetamine; (R)-(–)-N-2-propynylmethamphetamine |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a697046 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth, transdermal (patch) |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 10% (oral), 73% (patch) |
| Protein binding | Unknown[3] |
| Metabolism | Intestines and liver |
| Metabolites | N-Desmethylselegiline, levoamphetamine, levomethamphetamine |
| Elimination half-life | 1.5–3.5 hours (oral),[4] 18–25 hours (transdermal)[5] |
| Excretion | Urine |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.109.269 |
| Chemical and physical data | |
| Formula | C13H17N |
| Molar mass | 187.286 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Selegiline acts as a monoamine oxidase inhibitor, and increases levels of monoamine neurotransmitters in the brain. At typical clinical doses used for Parkinson's disease, selegiline is a selective and irreversible inhibitor of monoamine oxidase B (MAO-B), increasing levels of dopamine in the brain. In larger doses (more than 20 mg/day), it loses its specificity for MAO-B and also inhibits MAO-A, which increases serotonin and norepinephrine levels in the brain. In addition to its monoamine oxidase inhibition, selegiline is a prodrug of levomethamphetamine and levoamphetamine, which act as norepinephrine releasing agents and may also be involved in its effects and side effects.[6][7][6] These metabolites are greatly minimized with the oral disintegrating tablet and transdermal patch formulations of selegiline.[8][9]
The racemic form of selegiline, deprenyl, was discovered and studied in the early 1960s.[10][11] Subsequently, selegiline was purified from deprenyl and was itself studied and developed.[10] Selegiline was approved as an oral pill for the treatment of Parkinson's disease in 1989[12] and as a patch for the treatment of depression in 2006.[11] In addition to its medical use, there has been interest in selegiline as a potential anti-aging and longevity drug and as a nootropic or "smart drug".[13]
Medical uses
editParkinson's disease
editIn its pill form, selegiline is used to treat symptoms of Parkinson's disease.[14] It is most often used as an adjunct to drugs such as levodopa (L-DOPA), although it has been used off-label as a monotherapy.[15][16] The rationale for adding selegiline to levodopa is to decrease the required dose of levodopa and thus reduce the motor complications of levodopa therapy.[17] Selegiline delays the point when levodopa treatment becomes necessary from about 11 months to about 18 months after diagnosis.[18] There is some evidence that selegiline acts as a neuroprotectant and reduces the rate of disease progression, though this is disputed.[16][17]
Selegiline has also been used off-label as a palliative treatment for dementia in Alzheimer's disease.[16]
Depression
editSelegiline is also delivered via a transdermal patch used as a treatment for major depressive disorder.[19][20] Administration of transdermal selegiline bypasses hepatic first pass metabolism. This avoids inhibition of gastrointestinal and hepatic MAO-A activity, which would result in an increase of food-borne tyramine in the blood and possible related adverse effects, while allowing for a sufficient amount of selegiline to reach the brain for an antidepressant effect.[9]
A quantitative review published in 2015 found that for the pooled results of the pivotal trials, the number needed to treat (a sign of effect size, so a low number is better) for the patch for symptom reduction was 11, and for remission, was 9.[20] The number needed to harm (inverse of the NNT, a high number here is better) ranged from 387 for sexual side effects to 7 for application site reaction.[20] With regard to the likelihood to be helped or harmed (LHH), the analysis showed that the selegiline patch was 3.6 times as likely to lead to a remission vs. a discontinuation due to side effects; the LHH for remission vs. incidence of insomnia was 2.1; the LHH for remission vs. discontinuation due to insomnia was 32.7. The LHH for remission vs. insomnia and sexual dysfunction were both very low.[20]
Special populations
editFor all human uses and all forms, selegiline is pregnancy category C: studies in pregnant lab animals have shown adverse effects on the fetus but there are no adequate studies in humans.[14][19]
Available forms
editSelegiline is available in the form of 5 mg oral tablets and capsules (brand name Eldepryl and generics), 1.25 mg orally disintegrating tablets (brand name Zelapar), and 6 mg/24-hour, 9 mg/24-hour, and 12 mg/24-hour transdermal patches (brand name Emsam).[21]
Side effects
editSide effects of the tablet form in conjunction with levodopa include, in decreasing order of frequency, nausea, hallucinations, confusion, depression, loss of balance, insomnia, increased involuntary movements, agitation, slow or irregular heart rate, delusions, hypertension, new or increased angina pectoris, and syncope.[14] Most of the side effects are due to a high dopamine signaling, and can be alleviated by reducing the dose of levodopa.[1]
The main side effects of the patch form for depression include application-site reactions, insomnia, diarrhea, and sore throat.[19] The selegiline patch carries a black box warning about a possible increased risk of suicide, especially for young people,[19] as do all antidepressants since 2007.[22]
Interactions
editBoth the oral and patch forms come with strong warnings against combining selegiline with drugs that could produce serotonin syndrome, such as SSRIs and the cough medicine dextromethorphan.[14][19][23] Selegiline in combination with the opioid analgesic pethidine is not recommended, as it can lead to severe adverse effects.[23] Several other synthetic opioids such as tramadol and methadone, as well as various triptans, are contraindicated due to potential for serotonin syndrome.[24][25]
Birth control pills containing ethinylestradiol and a progestin increase the bioavailability of selegiline by 10- to 20-fold.[26] High levels can lead to loss of MAO-B selectivity, and selegiline may begin inhibiting MAO-A as well. This increases susceptibility to side effects of non-selective MAOIs, such as tyramine-induced hypertensive crisis and serotonin toxicity when combined with serotonergic medications.[26]
Both forms of the drug carry warnings about food restrictions to avoid hypertensive crisis that are associated with MAO inhibitors.[14][19] The patch form was created in part to overcome food restrictions; clinical trials showed that it was successful. Additionally, in post-marketing surveillance from April 2006 to October 2010, only 13 self-reports of possible hypertensive events or hypertension were made out of 29,141 exposures to the drug, and none were accompanied by objective clinical data.[20] The lowest dose of the patch method of delivery, 6 mg/24 hours, does not require any dietary restrictions.[27] Higher doses of the patch and oral formulations, whether in combination with the older non-selective MAOIs or in combination with the reversible MAO-A inhibitor moclobemide, require a low-tyramine diet.[23]
Selegiline "potently" or "strongly" inhibits the cytochrome P450 enzyme CYP2B6 in vitro.[28][29][30] It can inhibit the metabolism of bupropion, a major CYP2B6 substrate, into its active metabolite hydroxybupropion.[28][29][30] One small study observing three patients found that selegiline was safe in combination with bupropion.[30][31]
Pharmacology
editPharmacodynamics
editSelegiline is a selective inhibitor of MAO-B, irreversibly inhibiting it by binding to it covalently.[1][32] It is generally believed to exert its effects by blocking the breakdown of dopamine, thus increasing its activity;[33] however, recent evidence suggests that MAO-A is solely or almost entirely responsible for the metabolism of dopamine.[34] Its possible neuroprotective properties may be due to protecting nearby neurons from the free oxygen radicals that are released by MAO-B activity. At higher doses, selegiline loses its selectivity for MAO-B and inhibits MAO-A as well.[1] Selegiline has been called the "first synthetic catecholaminergic activity enhancer substance".[35][36]
Following a single 5 or 10 mg oral dose of selegiline, 86% to 90% of MAO-B activity in blood platelets was inhibited within 2 to 4 hours and 98% of activity was inhibited after 24 hours.[3][37] Inhibition of platelet MAO-B activity persisted at above 90% for 5 days and almost 14 days were required before activity returned to baseline.[3][37] A lower dose of selegiline of 1 mg/day also inhibits MAO-B activity by 70 to 100%.[37] However, despite this potent and long-lasting inhibition, optimal effectiveness in Parkinson's disease requires 10 mg/day and the effectiveness lasts only about 2 to 3 days with discontinuation of selegiline.[37] Taken together, selegiline almost completely inhibits platelet MAO-B act a dosage of 10 mg/day.[8] It is assumed that peripheral and brain MAO-B are inhibited with selegiline to very similar extents, although this is also not certain.[38][39][37]
In vitro, selegiline is about 1,000 times more potent in inhibiting platelet MAO-B relative to intestinal MAO-A.[37] The clinical selectivity of selegiline for MAO-B is lost at doses of the drug above 20 mg/day.[37] In a study of post-mortem individuals who took selegiline 10 mg/day for an average of 6 days prior to death, MAO-A activity in the brain was inhibited by 38 to 86%.[37][40] More recent studies have also found inhibition of brain MAO-A by 33 to 70% in humans.[41][42][43] However, while brain dopamine and phenethylamine levels are substantially increased (by 40–350% and 1,300%, respectively), brain serotonin and 5-hydroxyindoleacetic acid (5-HIAA) levels remain unchanged or 5-HIAA levels were only somewhat decreased.[40][37] It has been shown that MAO-A in the brain must be inhibited by nearly 85% before serotonin, norepinephrine, or dopamine levels increase and result in increased functional activity.[40] Selegiline at an oral dosage of 10 mg/day does not cause the "cheese effect" as assessed by oral tyramine and phenethylamine challenge tests.[3] These findings indicate that selegiline does not importantly inhibit MAO-A at a dosage of 10 mg/day.[3] However, a dosage of 20 mg/day selegiline did increase the pressor effect of tyramine, indicating that doses this high and above can significantly inhibit MAO-A.[40]
Besides increasing brain dopamine levels via MAO-B inhibition, selegiline strongly increases endogenous levels of β-phenethylamine (PEA), a major substrate of MAO-B.[37] Levels in of PEA in the brain are increased 10- to 30-fold and levels in urine are increased 20- to 90-fold.[37] PEA is normally present in small amounts in the brain and urine and has been referred to as "endogenous amphetamine".[37] Similarly to amphetamines, it induces the release of norepinephrine and dopamine and produces psychostimulant effects.[37] Selegiline also strongly increases levels of PEA with exogenous administration of PEA.[37] The increase in endogenous levels of PEA with selegiline might be involved in its effects, for instance claimed "psychic energizing" and mood-lifting effects.[13] In contrast to amphetamine psychostimulants however, selegiline has no misuse potential.[13]
Selegiline has potent pro-sexual effects in rodents, which are apparently not shared with other MAO-B inhibitors or clorgiline (an MAO-A inhibitor).[37] The pro-sexual effects are stronger than those of dopamine agonists like apomorphine and bromocriptine and high doses of amphetamine.[37] However, these pro-sexual effects in rodents were not subsequently confirmed in primates.[37]
Selegiline also inhibits the cytochrome P450 enzymes CYP2A6 and CYP2B6.[44][29] The CYP2A6 inhibition can increase the effects of nicotine.[44] Selegiline additionally appears to activate σ1 receptors, having a relatively high affinity for these receptors of approximately 400 nM.[45][46] The drug has a weak dopamine releasing effect, weakly blocks dopamine receptors, and weakly inhibits the reuptake of norepinephrine, but these actions are all of very low potency and are of questionable clinical significance.[37] Selegiline does not significantly inhibit the brain dopamine transporter in humans at clinical doses.[43]
Besides selegiline itself, its three major metabolites, desmethylselegiline, levomethamphetamine, and levoamphetamine, are pharmacologically active.[6] Compared to selegiline, desmethylselegiline is 60-fold less potent in inhibiting MAO-B in vitro, but is only 3- to 6-fold less potent in vivo.[3] Although desmethylselegiline levels with selegiline therapy are low, selegiline and desmethylselegiline are highly potent MAO-B inhibitors due to the irreversible nature of their inhibition.[37] As such, desmethylselegiline may contribute significantly to the MAO-B inhibition with selegiline.[37] Levomethamphetamine and levoamphetamine are sympathomimetic agents that work by inducing the release of norepinephrine.[6] They might improve fatigue, but can also produce cardiovascular side effects like increased heart rate and blood pressure and reportedly may be able to cause insomnia, psychiatric disturbances, and psychosis.[6][8] They are ten times less potent at stimulating locomotor activity (a measure of psychostimulant effect) than dextromethamphetamine and dextroamphetamine in rodents.[6][47] It is unknown what concentrations of levomethamphetamine and levoamphetamine produce sympathomimetic and other effects in humans and whether such concentrations are achieved with selegiline therapy.[6] However, cardiovascular side effects of selegiline have been found clinically and have been attributed to its amphetamine metabolites.[48][38] For comparison, rasagiline, which lacks amphetamine metabolites, has shown fewer adverse effects in clinical studies.[48][38][49]
It is unknown whether selegiline's amphetamine metabolites are involved in its effectiveness in the treatment of Parkinson's disease.[37] However, this has been deemed unlikely.[37] High doses of levoamphetamine, for instance 50 mg/day, have been reported to have some degree of effectiveness in the treatment of Parkinson's disease.[37] However, this could not be relevant to selegiline, which is administered at a dose of 10 mg/day.[37] Accordingly, relatively low levels of levoamphetamine occur with selegiline.[37] In one clinical study, levels of the amphetamine metabolites of selegiline were manipulated and there were no changes in clinical symptoms of Parkinson's disease.[37] This led the researchers to conclude that the beneficial clinical effects of selegiline in Parkinson's disease were not due to its amphetamine metabolites.[37] It is possible that there could be some small synergistic beneficial effect of selegiline with its amphetamine metabolites, but this has been considered improbable.[37] In any case, as previously mentioned, the amphetamine metabolites of selegiline might produce other effects, like reduced fatigue, as well as side effects.[6][8][48] Newer formulations of selegiline, such as the orally disintegrating tablet and the transdermal patch, have been developed which strongly reduce formation of the amphetamine metabolites and their associated effects.[8][9] In addition, other MAO-B inhibitors that do not metabolize into amphetamines, like rasagiline, have been developed and introduced.[6]
Pharmacokinetics
editAbsorption
editSelegiline has an oral bioavailability of about 10%.[3] The average time to peak levels of selegiline is 0.6 to 1.4 hours, with a range in one study of about 0.5 to 1.5 hours.[3]
The circulating levels of selegiline and its metabolites following a single 10 mg oral dose have been studied.[3] The metabolites of selegiline include desmethylselegiline, levomethamphetamine, and levoamphetamine.[3] The average peak concentrations of selegiline across several studies ranged from 0.84 ± 0.6 μg/L to 2.2 ± 1.2 μg/L and the AUC levels ranged from 1.26 ± 1.19 μg⋅h/L to 2.17 ± 2.59 μg⋅h/L.[3] In the case of desmethylselegiline, the time to peak has been reported to be 0.8 ± 0.2 hours, the peak levels were 7.84 ± 2.11 μg/L to 13.4 ± 3.2 μg/L, and the area-under-the-curve (AUC) levels were 15.05 ± 4.37 μg⋅h/L to 40.3 ± 10.7 μg⋅h/L.[3] For levomethamphetamine, the peak levels were 10.2 ± 1.5 μg/L and the AUC levels were 150.2 ± 21.6 μg⋅h/L, whereas for levoamphetamine, the peak levels were 3.6 ± 2.9 μg/L and the AUC levels were 61.7 ± 44.0 μg⋅h/L.[3] For comparison, following a single 10 mg oral dose of dextromethamphetamine or dextroamphetamine, peak levels of these agents have been reported to range from 14 to 90 μg/L and from 15 to 34 μg/L, respectively.[50] Levels of desmethylselegiline and levomethamphetamine are 10- to 20-fold higher than selegiline levels with oral selegiline therapy.[51]
With repeated administration of selegiline, there is an accumulation of selegiline and its metabolites.[3] With a dosage of 10 mg once a day or 5 mg twice daily, peak levels of selegiline were 1.59 ± 0.89 μg/L to 2.33 ± 1.76 μg/L and AUC levels of selegiline were 6.92 ± 5.39 μg⋅h/L to 7.84 ± 5.43 μg⋅h/L after 1 week of treatment.[3] This equated to a 1.9- to 2.6-fold accumulation in peak levels and a 3.6- to 5.5-fold accumulation in AUC levels.[3] The metabolites of selegiline accumulate to a smaller extent than selegiline.[3] The AUC levels of desmethylselegiline increased by 1.5-fold and the peak and AUC levels of levomethamphetamine and levoamphetamine increased by 2-fold following 1 week of treatment with selegiline.[3]
The oral bioavailability of selegiline increases when it is ingested together with a fatty meal, as the molecule is fat-soluble.[1][52] There is a 3-fold increase in peak levels of selegiline and a 5-fold increase in AUC levels when it is taken orally with food.[3] The elimination half-life of selegiline is unchanged when it is taken with food.[3] In contrast to selegiline itself, the pharmacokinetics of its metabolites, desmethylselegililne, levomethamphetamine, and levoamphetamine, are unchanged when selegiline is taken with food.[3]
A buccal formulation of selegiline was found to have 5-fold higher bioavailability, more consistent blood levels, and to produce fewer amphetamine metabolites than the standard oral tablet form.[53]
Distribution
editNo data were originally available on the plasma protein binding of selegiline.[3] It has been stated that the plasma protein binding of selegiline is 94%, but there is no actual evidence to support this figure.[3] Subsequent research found that its plasma protein binding is 89%.[9] The apparent volume of distribution of selegiline is 1,854 ± 824 L.[3] Selegiline and its metabolites cross the blood–brain barrier and enter the brain, where they most concentrated at the thalamus, basal ganglia, midbrain, and cingulate gyrus.[16][19] It especially accumulates in brain areas with high MAO-B content, such as the thalamus, striatum, cortex, and brainstem.[40] Concentrations of selegiline's metabolites in cerebrospinal fluid (CSF) are similar to those in blood, suggesting that accumulation in the brain does not occur.[40]
Metabolism
editSelegiline is mostly metabolized in the intestines and liver; it and its metabolites are excreted in the urine.[1] Selegiline is metabolized by cytochrome P450 to L-desmethylselegiline and levomethamphetamine.[54][55] Desmethylselegiline has some activity against MAO-B, but much less than that of selegiline.[33][32] It is thought to be further metabolized by CYP2C19.[56] Levomethamphetamine (the less potent of the two enantiomers of methamphetamine) is converted to levoamphetamine (the less potent of the two enantiomers of amphetamine, with regards to psychological effects).
Due to the presence of these metabolites, people taking selegiline may test positive for "amphetamine" or "methamphetamine" on drug screening tests.[57] While the amphetamine metabolites may contribute to selegiline's ability to inhibit reuptake of the neurotransmitters dopamine and norepinephrine, they have also been associated with orthostatic hypotension and hallucinations.[55][58][7] The recovery of selegiline from urine is high at 87%, which has caused some researchers to question the clinical relevance of its amphetamine metabolites.[59] The amphetamine metabolites are hydroxylated and, in phase II, conjugated by glucuronyltransferase.
The cytochrome P450 enzymes CYP2B6, CYP2C9, and CYP3A are thought to be significantly involved in the metabolism of selegiline on the basis of in vitro studies.[9][60][61] Other cytochrome P450 enzymes, including CYP1A2, CYP2A6, CYP2C8, CYP2D6, CYP2C19, and CYP2E1, may also be involved.[9][62][60][61] One review concluded that CYP2B6 and CYP2C19 are the leading candidates in selegiline metabolism.[63] CYP2B6 is thought to N-demethylate selegiline into desmethylselegiline and CYP2B6 and CYP2C19 are thought to N-depropargylate selegiline into levomethamphetamine.[9][60] Additionally, CYP2B6 and CYP2C19 are thought to metabolize desmethylselegiline into levoamphetamine and CYP2B6 is thought to N-demethylate levomethamphetamine into levoamphetamine.[9][60] CYP2D6 and CYP2C19 metabolizer phenotypes did not significantly affect the pharmacokinetics of selegiline, suggesting that these enzymes are minimally involved in its metabolism.[63][61][51] However, although most pharmacokinetic variables were unaffected, AUC levels of levomethamphetamine were 46% higher in CYP2D6 poor metabolizers compared to extensive metabolizers and desmethylselegiline AUC levels were 68% in CYP2C19 poor metabolizers compared to extensive metabolizers.[61][51] As with CYP2D6 and CYP2C19, CYP3A4 and CYP3A5 are unlikely to be majorly involved in the metabolism of selegiline as the strong inhibitor itraconazole has minimal impact on its pharmacokinetics.[63][64] Besides its major metabolites of desmethylselegiline, levomethamphetamine, and levoamphetamine, selegiline-N-oxide and formaldehyde are formed.[65] In addition, more than 40 minor metabolites of selegiline have been either detected or proposed.[65]
The oral clearance of selegiline is 59.4 ± 43.7 L/min.[3] This is described as very high and as almost 30-fold higher than hepatic blood flow.[3] The renal clearance of selegiline is 0.0072 L/h and is very low compared to its oral clearance.[3] These findings suggest that selegiline is extensively metabolized not only by the liver but also by non-hepatic tissues.[3]
A newer anti-Parkinson MAO-B inhibitor, rasagiline, metabolizes into 1(R)-aminoindan, which has no amphetamine-like characteristics.[66]
Elimination
editThe average elimination half-life of selegiline after a single oral dose ranges from 1.2 hours to 1.9 hours across studies.[3] With repeated administration, the half-life of selegiline increases to 7.7 ± 12.6 hours to 9.6 ± 13.6 hours.[3] The elimination half-life of selegiline's metabolite, desmethylselegiline, has been reported to range from 2.2 ± 0.6 hours to 3.8 hours.[3] The half-lives of its metabolites levomethamphetamine and levoamphetamine have been reported to be 14 hours and 16 hours, respectively.[3] Following repeated administration, the half-life of desmethylselegiline increased from 3.8 hours with the first dose to 9.5 hours following 1 week of daily selegiline doses.[3]
Approximately 0.01% to 0.03% unchanged selegiline is excreted in urine with an oral dose.[3] This indicates that selegiline is essentially completely metabolized prior to excretion.[3]
Orally disintegrating tablet
editSelegiline as an orally disintegrating tablet (ODT) achieves blood levels of selegiline at a dose of 1.25 mg/day that are similar to those with conventional oral selegiline at a dose of 10 mg/day.[8] In addition, there is an at least 90% reduction in metabolites of selegiline including desmethylselegiline, levomethamphetamine, and levoamphetamine with the ODT formulation of selegiline compared to conventional oral selegiline.[8] Hence, levels of these metabolites are 10-fold lower with the ODT formulation.[48] This formulation of selegiline retains selectivity for MAO-B over MAO-A and likewise does not cause the "cheese effect" with consumption of tyramine-rich foods.[8]
Transdermal patch
editFollowing application of the patch to humans, an average of 25% to 30% of the selegiline content is delivered systemically over 24 hours.[9] This equates to about 0.3 mg selegiline per cm2 over 24 hours.[9] Transdermal dosing results in significantly higher exposure to selegiline and lower exposure to all metabolites when compared to oral dosing.[9] Selegiline levels are 50-fold higher and exposure to metabolites 70% lower with transdermal administration compared to oral administration at equivalent doses.[9] The patch has approximately 75% bioavailability, compared to 10% with the conventional oral form.[9] These differences are due to the extensive first-pass metabolism of the pill form and low first-pass metabolism of the patch form. The site of application is not a significant factor in how the drug is distributed. In humans, selegiline does not accumulate in the skin, nor is it metabolized there.[19]
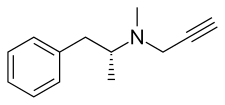
Chemistry
editSelegiline belongs to the phenethylamine and amphetamine chemical families. It is also known as L-deprenyl, as well as (R)-(–)-N,α-dimethyl-N-(2-propynyl)phenethylamine or (R)-(–)-N-methyl-N-2-propynylamphetamine. The compound is a derivative of levomethamphetamine (L-methamphetamine) with a propargyl group attached to the nitrogen atom. It is also known as N-propargyl-L-methamphetamine.[8] This detail is borrowed from pargyline, an older MAO-B inhibitor of the phenylalkylamine group.[35] Selegiline is the levorotatory enantiomer of the racemic mixture deprenyl.
Selegiline is synthesized by the alkylation of (–)-methamphetamine using propargyl bromide.[67][68][69][70]
Another clinically used MAOI of the amphetamine class is tranylcypromine.
History
editFollowing the discovery in 1952 that the tuberculosis drug iproniazid elevated the mood of people taking it, and the subsequent discovery that the effect was likely due to inhibition of MAO, many people and companies started trying to discover MAO inhibitors to use as antidepressants. Deprenyl (the racemic form of selegiline) was discovered by Zoltan Ecseri at the Hungarian drug company, Chinoin (part of Sanofi since 1993),[71] which they called E-250.[72]: 66–67 Chinoin received a patent on the drug in 1962 and the compound was first published in the scientific literature in English in 1965.[72]: 67 [73] Work on the biology and effects of E-250 in animals and humans was conducted by a group led by József Knoll at Semmelweis University which was also in Budapest.[72]: 67
Deprenyl is a racemic compound (a mixture of two isomers called enantiomers). Further work determined that the levorotatory enantiomer was a more potent MAO-inhibitor, which was published in 1967, and subsequent work was done with the single enantiomer L-deprenyl.[72]: 67 [74][75]
In 1971, Knoll showed that selegiline selectively inhibits the B-isoform of monoamine oxidase (MAO-B) and proposed that it is unlikely to cause the infamous "cheese effect" (hypertensive crisis resulting from consuming foods containing tyramine) that occurs with non-selective MAO inhibitors. A few years later, two Parkinson's disease researchers based in Vienna, Peter Riederer and Walther Birkmayer, realized that selegiline could be useful in Parkinson's disease. One of their colleagues, Prof. Moussa B.H. Youdim, visited Knoll in Budapest and took selegiline from him to Vienna. In 1975, Birkmayer's group published the first paper on the effect of selegiline in Parkinson's disease.[75][76]
In the 1970s there was speculation that it could be useful as an anti-aging drug or aphrodisiac.[77]
In 1987 Somerset Pharmaceuticals in New Jersey, which had acquired the US rights to develop selegiline, filed a new drug application (NDA) with the FDA to market the drug for Parkinson's disease in the US.[12] While the NDA was under review, Somerset was acquired in a joint venture by two generic drug companies, Mylan and Bolan Pharmaceuticals.[12] Selegiline was approved for Parkinson's disease by the FDA in 1989.[12]
In the 1990s, J. Alexander Bodkin at McLean Hospital, an affiliate of Harvard Medical School, began a collaboration with Somerset to develop delivery of selegiline via a transdermal patch in order to avoid the well known dietary restrictions of MAO inhibitors.[77][78][79] Somerset obtained FDA approval to market the patch in 2006.[80]
Non-medical use
editAnti-aging and longevity
editJoseph Knoll and his team are credited with having developed selegiline. Although selegeline's development as a potential treatment for Parkinson's, Alzheimer's, and major depressive disorder was headed by other teams, Knoll remained at the forefront of research into the potential longevity enhancing effects of selegiline up until his death in 2018.[81][82][83] Knoll published How Selegiline ((-)-Deprenyl) Slows Brain Aging (2018) wherein he claims that:
"In humans, maintenance from sexual maturity on (-)-deprenyl (1mg daily) is, for the time being, the most promising prophylactic treatment to fight against the age related decay of behavioral performances, prolonging life, and preventing or delaying the onset of age-related neurodegenerative diseases such as Parkinson's and Alzheimer's".[84]
The mechanism of selegiline's longevity-promoting effect has been researched by several groups, including Joseph Knoll and his associates at Semmelweis University, Budapest.[11] The drug has been determined to be a catecholaminergic activity enhancer when present in minuscule concentrations far below those at which monoamine oxidase inhibitory activity can be observed, thereby potentiating the release of catecholamine neurotransmitters in response to stimuli. Knoll maintains that micro-doses of selegiline act as a synthetic analogue to a known or unknown trace amine in order to preserve the brain catecholaminergic system, which he perceives as integral to the organism's ability to function in an adaptive, goal-directed and motivated manner during advancing physical age:
" ... enhancer regulation in the catecholaminergic brain stem neurons play[s] a key role in controlling the uphill period of life and the transition from adolescence to adulthood. The results of our longevity studies support the hypothesis that quality and duration of life rests upon the inborn efficiency of the catcholaminergic brain machinery, i.e. a high performing, long-living individual has a more active, more slowly deteriorating catecholaminergic system than its low performing, shorter living peer. Thus, a better brain engine allows for a better performance and a longer lifespan.
...
Since the catecholaminergic and serotonergic neurons in the brain stem are of key importance in ensuring that the mammalian organism works as a purposeful, motivated, goal-directed entity, it is hard to overestimate the significance of finding safe and efficient means to slow the decay of these systems with passing time. The conclusion that the maintenance on (-)-deprenyl that keeps the catecholaminergic neurnsn a higher activity level is a safe and efficient anti- aging therapy follows from the discovery of the enhancer regulation in the catecholaminergic neurons of the brain stem. From the finding that this regulation starts working on a high activity level after weaning and the enhanced activity subsists during the uphill period of life, until sexual hormones dampen the enhancer regulation in the catecholaminergic and serotonergic neurons in the brain stem, and this event signifies the transition from developmental longevity into postdevelopmental longevity, the downhill period of life."[84]
Nootropic or "smart drug"
editSelegiline is considered by some to be a nootropic,[85] both at clinical and sub-clinical dosages, and has been used off-label to improve cognitive performance. It has been shown to have protective activity against a range of neurotoxins and to increase the production of several brain growth factors, such as nerve growth factor, brain-derived neurotrophic factor, and glial cell-derived neurotrophic factor.[11] It has been demonstrated in numerous animal models to improve learning ability and preserve it during both ischemia and aging.[86][87][88][89]
Society and culture
editIn E for Ecstasy (a book examining the uses of the street drug ecstasy in the UK) the writer, activist and ecstasy advocate Nicholas Saunders highlighted test results showing that certain consignments of the drug also contained selegiline.[90] Consignments of ecstasy known as "Strawberry" contained what Saunders described as a "potentially dangerous combination of ketamine, ephedrine and selegiline," as did a consignment of "Sitting Duck" Ecstasy tablets.[91]
David Pearce wrote The Hedonistic Imperative[92] six weeks after starting taking selegiline.[93]
In Gregg Hurwitz's novel Out of the Dark,[94] selegiline (Emsam) and tyramine-containing food were used to assassinate the president of the United States.
Veterinary use
editIn veterinary medicine, selegiline is sold under the brand name Anipryl (manufactured by Zoetis). It is used in dogs to treat canine cognitive dysfunction and, at higher doses, pituitary-dependent hyperadrenocorticism (PDH).[95][96] Canine cognitive dysfunction is a form of dementia that mimics Alzheimer's disease in humans. Geriatric dogs treated with selegiline show improvements in sleeping pattern, reduced incontinence, and increased activity level; most show improvements by one month.[97][98] Though it is labeled for dog use only, selegiline has been used off-label for geriatric cats with cognitive dysfunction.[99]
Selegiline's efficacy in treating pituitary-dependent hyperadrenocorticism has been disputed.[95] Theoretically, it works by increasing dopamine levels, which downregulates the release of ACTH, eventually leading to reduced levels of cortisol.[99] Some claim that selegiline is only effective at treating PDH caused by lesions in the anterior pituitary (which comprise most canine cases).[100] The greatest sign of improvement is lessening of abdominal distention.[97]
Side effects in dogs are uncommon, but they include vomiting, diarrhea, diminished hearing, salivation, decreased weight and behavioral changes such as hyperactivity, listlessness, disorientation, and repetitive motions.[96][100]
Selegiline does not appear to have a clinical effect on horses.[100]
Research
editADHD
editSelegiline has been limitedly studied in the treatment of attention deficit hyperactivity disorder (ADHD) in both children/adolescents and adults.[101][102] In a small randomized trial of selegiline for treatment of ADHD in children, there were improvements in attention, hyperactivity, and learning/memory performance but not in impulsivity.[103] A small clinical randomized trial compared selegiline to methylphenidate, a first line treatment for ADHD, and reported equivalent efficacy as assessed by parent and teacher ratings.[104] In another small randomized controlled trial of selegiline for the treatment of adult ADHD, a high dose of the medication for 6 weeks was not significantly more effective than placebo in improving symptoms.[102][105][106]
Motivational disorders
editSelegiline has been found to increase effort expenditure and to reverse pharmacologically-induced motivational deficits in rodents.[107][108][109] In case reports and small clinical studies, selegiline has been reported to improve apathy in people with traumatic brain injury and schizophrenia.[107] However, no evidence of benefit was found with selegiline in people with Alzheimer's disease.[107]
References
edit- ^ a b c d e f g "Selegiline". Drugs.com. Retrieved February 7, 2016.
- ^ Anvisa (March 31, 2023). "RDC Nº 784 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 784 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published April 4, 2023). Archived from the original on August 3, 2023. Retrieved August 16, 2023.
- ^ a b c d e f g h i j k l m n o p q r s t u v w x y z aa ab ac ad ae af ag ah ai Mahmood I (August 1997). "Clinical pharmacokinetics and pharmacodynamics of selegiline. An update". Clin Pharmacokinet. 33 (2): 91–102. doi:10.2165/00003088-199733020-00002. PMID 9260033.
- ^ "Résumé des Caractéristiques du Produit".
- ^ https://www.accessdata.fda.gov/drugsatfda_docs/label/2007/021336s002lbl.pdf [bare URL PDF]
- ^ a b c d e f g h i Gerlach, Manfred; Reichmann, Heinz; Riederer, Peter (2012). "A critical review of evidence for preclinical differences between rasagiline and selegiline". Basal Ganglia. 2 (4): S9–S15. doi:10.1016/j.baga.2012.04.032.
- ^ a b Yasar S, Goldberg JP, Goldberg SR (January 1, 1996). "Are metabolites of l-deprenyl (Selegiline) useful or harmful? Indications from preclinical research". Deprenyl — Past and Future. Vol. 48. pp. 61–73. doi:10.1007/978-3-7091-7494-4_6. ISBN 978-3-211-82891-5. PMID 8988462.
{{cite book}}:|journal=ignored (help) - ^ a b c d e f g h i Poston KL, Waters C (October 2007). "Zydis selegiline in the management of Parkinson's disease". Expert Opin Pharmacother. 8 (15): 2615–24. doi:10.1517/14656566.8.15.2615. PMID 17931095.
- ^ a b c d e f g h i j k l m Lee KC, Chen JJ (November 2007). "Transdermal selegiline for the treatment of major depressive disorder". Neuropsychiatric Disease and Treatment. 3 (5): 527–537. PMC 2656289. PMID 19300583.
- ^ a b Parnham, M. J. (1993). "The History of l-Deprenyl". Inhibitors of Monoamine Oxidase B. Milestones in Drug Therapy (in German). Basel: Birkhäuser Basel. pp. 237–251. doi:10.1007/978-3-0348-6348-3_12. ISBN 978-3-0348-6349-0.
- ^ a b c d Miklya I (November 2016). "The significance of selegiline/(-)-deprenyl after 50 years in research and therapy (1965-2015)". Molecular Psychiatry. 21 (11): 1499–1503. doi:10.1038/mp.2016.127. PMID 27480491.
- ^ a b c d Seaman J, Landry JT (2011). Mylan: 50 Years of Unconventional Success: Making Quality Medicine Affordable and Accessible. University Press of New England. p. 50. ISBN 978-1-61168-269-4.
- ^ a b c Schneider LS, Tariot PN, Goldstein B (December 1994). "Therapy with l-deprenyl (selegiline) and relation to abuse liability". Clin Pharmacol Ther. 56 (6 Pt 2): 750–6. doi:10.1038/clpt.1994.205. PMID 7995017.
Some studies in patients with PD suggest that l-deprenyl might have relatively nonspecific ameliorative effects on mood and arousal.32,33 Indeed, the original reports in mixed psychiatric populations suggested a "psychic energizing" effect of the drug.34 Conceivably, such effects of l-deprenyl might be mediated through an increase in endogenous β-phenethylamine (β-PEA) consequent to MAO-B inhibition. β-PEA is a recognized psychomotor stimulant drug and, as higher levels develop in the presence of l-deprenyl, significant effects on mood might be anticipated (e.g., Greenshaw et al.35 and Timar and Knoll36).
- ^ a b c d e Selegiline oral label. Updated December 31, 2008
- ^ Riederer P, Lachenmayer L, Laux G (August 2004). "Clinical applications of MAO-inhibitors". Current Medicinal Chemistry. 11 (15): 2033–2043. doi:10.2174/0929867043364775 (inactive April 2, 2024). PMID 15279566.
{{cite journal}}: CS1 maint: DOI inactive as of April 2024 (link) - ^ a b c d "Selegiline Hydrochloride Monograph for Professionals". Drugs.com. Retrieved February 23, 2018.
- ^ a b Ives NJ, Stowe RL, Marro J, Counsell C, Macleod A, Clarke CE, et al. (September 2004). "Monoamine oxidase type B inhibitors in early Parkinson's disease: meta-analysis of 17 randomised trials involving 3525 patients". BMJ. 329 (7466): 593. doi:10.1136/bmj.38184.606169.AE. PMC 516655. PMID 15310558.
- ^ Riederer P, Lachenmayer L (November 2003). "Selegiline's neuroprotective capacity revisited". Journal of Neural Transmission. 110 (11): 1273–1278. doi:10.1007/s00702-003-0083-x. PMID 14628191. S2CID 20232921.
- ^ a b c d e f g h Emsam label Last revised Sept 2014. Index page at FDA
- ^ a b c d e Citrome L, Goldberg JF, Portland KB (November 2013). "Placing transdermal selegiline for major depressive disorder into clinical context: number needed to treat, number needed to harm, and likelihood to be helped or harmed". Journal of Affective Disorders. 151 (2): 409–417. doi:10.1016/j.jad.2013.06.027. PMID 23890583.
- ^ "Drugs@FDA: FDA-Approved Drugs". accessdata.fda.gov. Retrieved July 1, 2024.
- ^ Friedman RA, Leon AC (June 2007). "Expanding the black box - depression, antidepressants, and the risk of suicide". The New England Journal of Medicine. 356 (23): 2343–2346. doi:10.1056/NEJMp078015. PMID 17485726.
- ^ a b c Heinonen EH, Myllylä V (July 1998). "Safety of selegiline (deprenyl) in the treatment of Parkinson's disease". Drug Safety. 19 (1): 11–22. doi:10.2165/00002018-199819010-00002. PMID 9673855. S2CID 9632549.
- ^ Csoti I, Storch A, Müller W, Jost WH (December 1, 2012). "Drug interactions with selegiline versus rasagiline". Basal Ganglia. Monoamine oxidase B Inhibitors. 2 (4, Supplement): S27–S31. doi:10.1016/j.baga.2012.06.003. ISSN 2210-5336.
- ^ Gillman PK (October 2005). "Monoamine oxidase inhibitors, opioid analgesics and serotonin toxicity". British Journal of Anaesthesia. 95 (4): 434–441. doi:10.1093/bja/aei210. PMID 16051647.
- ^ a b Laine K, Anttila M, Helminen A, Karnani H, Huupponen R (March 1999). "Dose linearity study of selegiline pharmacokinetics after oral administration: evidence for strong drug interaction with female sex steroids". Br J Clin Pharmacol. 47 (3): 249–54. doi:10.1046/j.1365-2125.1999.00891.x. PMC 2014223. PMID 10215747.
- ^ Jessen L, Kovalick LJ, Azzaro AJ (April 2008). "The selegiline transdermal system (emsam): a therapeutic option for the treatment of major depressive disorder". P & T. 33 (4): 212–246. PMC 2730099. PMID 19750165.
- ^ a b Hedrich WD, Hassan HE, Wang H (September 2016). "Insights into CYP2B6-mediated drug-drug interactions". Acta Pharm Sin B. 6 (5): 413–425. doi:10.1016/j.apsb.2016.07.016. PMC 5045548. PMID 27709010.
Selegiline is frequently used in the treatment of Parkinson's disease. Sridar et al.178 have shown selegiline to be a strong inhibitor of CYP2B6-mediated metabolism of bupropion in vitro, increasing the Km of bupropion from 10 to 92 mmol/L and decreasing the kcat by approximately 50%178. This strong inhibition of CYP2B6 by selegiline highlights a serious potential of DDI for combination therapies involving bupropion.
- ^ a b c Zanger UM, Klein K (2013). "Pharmacogenetics of cytochrome P450 2B6 (CYP2B6): advances on polymorphisms, mechanisms, and clinical relevance". Front Genet. 4: 24. doi:10.3389/fgene.2013.00024. PMC 3588594. PMID 23467454.
- ^ a b c Sridar C, Kenaan C, Hollenberg PF (December 2012). "Inhibition of bupropion metabolism by selegiline: mechanism-based inactivation of human CYP2B6 and characterization of glutathione and peptide adducts". Drug Metab Dispos. 40 (12): 2256–66. doi:10.1124/dmd.112.046979. PMC 3500550. PMID 22936314.
- ^ Ritter JL, Alexander B (March 1997). "Retrospective study of selegiline-antidepressant drug interactions and a review of the literature". Ann Clin Psychiatry. 9 (1): 7–13. doi:10.1023/a:1026222106851. PMID 9167831.
- ^ a b Factor SA, Weiner W (2007). Parkinson's Disease: Diagnosis & Clinical Management (2nd ed.). Demos Medical Publishing. pp. 503, 505. ISBN 978-1-934559-87-1.
- ^ a b Katzung BG (2004). Basic and Clinical Pharmacology (9th ed.). Lange Medical Books/McGraw Hill. pp. 453. ISBN 978-0-07-141092-2.
- ^ Cho HU, Kim S, Sim J, Yang S, An H, Nam MH, et al. (July 2021). "Redefining differential roles of MAO-A in dopamine degradation and MAO-B in tonic GABA synthesis". Experimental & Molecular Medicine. 53 (7): 1148–1158. doi:10.1038/s12276-021-00646-3. PMC 8333267. PMID 34244591.
- ^ a b Miklya I (March 13, 2014). "The History of Selegiline/(-)-Deprenyl the First Selective Inhibitor of B-Type Monoamine Oxidase and The First Synthetic Catecholaminergic Activity Enhancer Substance". International Network for the History of Neuropsychopharmacology. Archived from the original on February 7, 2016. Retrieved January 7, 2016.
- ^ Knoll J (1997). "[History of deprenyl--the first selective inhibitor of monoamine oxidase type B]". Voprosy Meditsinskoi Khimii. 43 (6): 482–493. PMID 9503565.
- ^ a b c d e f g h i j k l m n o p q r s t u v w x y z aa ab Heinonen EH, Lammintausta R (1991). "A review of the pharmacology of selegiline". Acta Neurol Scand Suppl. 136: 44–59. doi:10.1111/j.1600-0404.1991.tb05020.x. PMID 1686954.
- ^ a b c Finberg JP (April 2019). "Inhibitors of MAO-B and COMT: their effects on brain dopamine levels and uses in Parkinson's disease". J Neural Transm (Vienna). 126 (4): 433–448. doi:10.1007/s00702-018-1952-7. PMID 30386930.
- ^ Finberg JP, Gillman K (2011). "Selective inhibitors of monoamine oxidase type B and the "cheese effect"". Int Rev Neurobiol. International Review of Neurobiology. 100: 169–90. doi:10.1016/B978-0-12-386467-3.00009-1. ISBN 978-0-12-386467-3. PMID 21971008.
As these propargyl inhibitors efficiently pass the blood–brain barrier, it may be assumed that the extent of MAO-B inhibition for a given dose in the brain is very close to that in the periphery, and animal experiments show this to be true (see Waldmeieret al., 1981).
- ^ a b c d e f Gerlach M, Youdim MB, Riederer P (December 1996). "Pharmacology of selegiline". Neurology. 47 (6 Suppl 3): S137–45. doi:10.1212/wnl.47.6_suppl_3.137s. PMID 8959982.
- ^ Tábi T, Vécsei L, Youdim MB, Riederer P, Szökő É (May 2020). "Selegiline: a molecule with innovative potential". J Neural Transm (Vienna). 127 (5): 831–842. doi:10.1007/s00702-019-02082-0. PMC 7242272. PMID 31562557.
- ^ Bartl J, Müller T, Grünblatt E, Gerlach M, Riederer P (April 2014). "Chronic monoamine oxidase-B inhibitor treatment blocks monoamine oxidase-A enzyme activity". J Neural Transm (Vienna). 121 (4): 379–83. doi:10.1007/s00702-013-1120-z. PMID 24272680.
- ^ a b Fowler JS, Logan J, Volkow ND, Shumay E, McCall-Perez F, Jayne M, Wang GJ, Alexoff DL, Apelskog-Torres K, Hubbard B, Carter P, King P, Fahn S, Gilmor M, Telang F, Shea C, Xu Y, Muench L (February 2015). "Evidence that formulations of the selective MAO-B inhibitor, selegiline, which bypass first-pass metabolism, also inhibit MAO-A in the human brain". Neuropsychopharmacology. 40 (3): 650–7. doi:10.1038/npp.2014.214. PMC 4289953. PMID 25249059.
- ^ a b Siu EC, Tyndale RF (March 2008). "Selegiline is a mechanism-based inactivator of CYP2A6 inhibiting nicotine metabolism in humans and mice". The Journal of Pharmacology and Experimental Therapeutics. 324 (3): 992–999. doi:10.1124/jpet.107.133900. PMID 18065502.
- ^ Itzhak Y (1994). Sigma Receptors. Academic Press. p. 84. ISBN 978-0-12-376350-1.
- ^ Stone TW (1993). Acetylcholine, Sigma Receptors, CCK and Eicosanoids, Neurotoxins. Taylor & Francis. p. 124. ISBN 978-0-7484-0063-8.
- ^ Taylor KM, Snyder SH (June 1970). "Amphetamine: differentiation by d and l isomers of behavior involving brain norepinephrine or dopamine". Science. 168 (3938): 1487–1489. doi:10.1126/science.168.3938.1487. PMID 5463064.
- ^ a b c d Fernandez HH, Chen JJ (December 2007). "Monoamine oxidase-B inhibition in the treatment of Parkinson's disease". Pharmacotherapy. 27 (12 Pt 2): 174S–185S. doi:10.1592/phco.27.12part2.174S. PMID 18041937.
These safety issues have been attributed in part to the amphetamine metabolites of selegiline and potential cardiovascular effects.50 [...] Cardiovascular adverse events in these studies may be attributed at least in part to a toxic effect of selegiline's amphetamine metabolites.
- ^ HoffmannDimpfel2013Müller T, Hoffmann JA, Dimpfel W, Oehlwein C (May 2013). "Switch from selegiline to rasagiline is beneficial in patients with Parkinson's disease". J Neural Transm (Vienna). 120 (5): 761–5. doi:10.1007/s00702-012-0927-3. PMID 23196982.
- ^ Paz-Ramos MI, Cruz SL, Violante-Soria V (2023). "Amphetamine-type Stimulants: Novel Insights into their Actions and use Patterns". Rev Invest Clin. 75 (3): 143–157. doi:10.24875/RIC.23000110. PMID 37441770.
- ^ a b c Laine K, Anttila M, Nyman L, Wahlberg A, Bertilsson L (May 2001). "CYP2C19 polymorphism is not important for the in vivo metabolism of selegiline". Eur J Clin Pharmacol. 57 (2): 137–42. doi:10.1007/s002280100289. PMID 11417445.
Selegiline is metabolised to two known metabolites, desmethylselegiline and I-methamphetamine (Fig. l) [7]. The oral bioavailability of selegiline is less than 10% due to extensive first-pass metabolism [8] and the concen- trations of these two metabolites are IO- to 20-fold higher in serum compared with the parent compound [6, 9].
- ^ Barrett JS, Szego P, Rohatagi S, Morales RJ, De Witt KE, Rajewski G, Ireland J (October 1996). "Absorption and presystemic metabolism of selegiline hydrochloride at different regions in the gastrointestinal tract in healthy males". Pharmaceutical Research. 13 (10): 1535–1540. doi:10.1023/A:1016035730754. PMID 8899847. S2CID 24654277.
- ^ Clarke A, Brewer F, Johnson ES, Mallard N, Hartig F, Taylor S, Corn TH (November 2003). "A new formulation of selegiline: improved bioavailability and selectivity for MAO-B inhibition". Journal of Neural Transmission. 110 (11): 1241–1255. doi:10.1007/s00702-003-0036-4. PMID 14628189. S2CID 711419.
- ^ Engberg G, Elebring T, Nissbrandt H (November 1991). "Deprenyl (selegiline), a selective MAO-B inhibitor with active metabolites; effects on locomotor activity, dopaminergic neurotransmission and firing rate of nigral dopamine neurons". The Journal of Pharmacology and Experimental Therapeutics. 259 (2): 841–847. PMID 1658311.
- ^ a b Lemke TL, Williams DA, eds. (2012). Foye's Principles of Medicinal Chemistry. Lippincott Williams & Wilkins. ISBN 978-1609133450.
- ^ Taavitsainen P, Anttila M, Nyman L, Karnani H, Salonen JS, Pelkonen O (May 2000). "Selegiline metabolism and cytochrome P450 enzymes: in vitro study in human liver microsomes". Pharmacology & Toxicology. 86 (5): 215–221. doi:10.1034/j.1600-0773.2000.pto860504.x. PMID 10862503.
- ^ Romberg RW, Needleman SB, Snyder JJ, Greedan A (November 1995). "Methamphetamine and amphetamine derived from the metabolism of selegiline". Journal of Forensic Sciences. 40 (6): 1100–1102. doi:10.1520/JFS13885J. PMID 8522918.
- ^ Bar Am O, Amit T, Youdim MB (January 2004). "Contrasting neuroprotective and neurotoxic actions of respective metabolites of anti-Parkinson drugs rasagiline and selegiline". Neuroscience Letters. 355 (3): 169–172. doi:10.1016/j.neulet.2003.10.067. PMID 14732458. S2CID 20471004.
- ^ Heinonen EH, Myllylä V, Sotaniemi K, Lamintausta R, Salonen JS, Anttila M, et al. (November 1989). "Pharmacokinetics and metabolism of selegiline". Acta Neurologica Scandinavica. Supplementum. 126: 93–99. doi:10.1111/j.1600-0404.1989.tb01788.x. PMID 2515726. S2CID 221440315.
- ^ a b c d Hidestrand M, Oscarson M, Salonen JS, Nyman L, Pelkonen O, Turpeinen M, Ingelman-Sundberg M (November 2001). "CYP2B6 and CYP2C19 as the major enzymes responsible for the metabolism of selegiline, a drug used in the treatment of Parkinson's disease, as revealed from experiments with recombinant enzymes". Drug Metab Dispos. 29 (11): 1480–4. PMID 11602525.
- ^ a b c d Kraemer T, Maurer HH (April 2002). "Toxicokinetics of amphetamines: metabolism and toxicokinetic data of designer drugs, amphetamine, methamphetamine, and their N-alkyl derivatives". Ther Drug Monit. 24 (2): 277–89. doi:10.1097/00007691-200204000-00009. PMID 11897973.
- ^ Magyar K (2011). "The pharmacology of selegiline". Int Rev Neurobiol. International Review of Neurobiology. 100: 65–84. doi:10.1016/B978-0-12-386467-3.00004-2. ISBN 978-0-12-386467-3. PMID 21971003.
- ^ a b c Rodrigues AD (June 2022). "Drug Interactions Involving 17α-Ethinylestradiol: Considerations Beyond Cytochrome P450 3A Induction and Inhibition". Clin Pharmacol Ther. 111 (6): 1212–1221. doi:10.1002/cpt.2383. PMID 34342002.
Unfortunately, available in vitro data sets for selegiline are confusing and do not necessarily shed light on the observed DDI with EE-containing OC. Selegiline is metabolized to two major metabolites (desmethylselegiline and methamphetamine) and panels of recombinant proteins present CYP2B6 and CYP2C19 as leading candidates, although metabolism is also reported for other CYP forms.50-52 Importantly, clinical data do not support the CYP3A4/5-dependent metabolism of selegiline, since itraconazole has a minimal impact on its PK.53 Likewise, both CYP2D6 and CYP2C19 can be ruled out because selegiline PK is not significantly impacted in poor metabolizer subjects.54,55 Although liver CYP2B6 is described as a major selegiline-metabolizing CYP in vitro, its expression in gut is low (Table S1)56,57, and there are no reports describing the PK of selegiline in CYP2B6 genotyped subjects.
- ^ Kivistö KT, Wang JS, Backman JT, Nyman L, Taavitsainen P, Anttila M, Neuvonen PJ (April 2001). "Selegiline pharmacokinetics are unaffected by the CYP3A4 inhibitor itraconazole". Eur J Clin Pharmacol. 57 (1): 37–42. doi:10.1007/s002280100278. PMID 11372588.
- ^ a b Kalász H, Magyar K, Szőke É, Adeghate E, Adem A, Hasan MY, Nurulain SM, Tekes K (2014). "Metabolism of selegiline [(-)-deprenyl)]". Curr Med Chem. 21 (13): 1522–30. doi:10.2174/0929867321666131218094352. PMID 24350849.
- ^ Chen JJ, Swope DM (August 2005). "Clinical pharmacology of rasagiline: a novel, second-generation propargylamine for the treatment of Parkinson disease". Journal of Clinical Pharmacology. 45 (8): 878–894. doi:10.1177/0091270005277935. PMID 16027398. S2CID 24350277. Archived from the original on July 11, 2012.
- ^ DE 1568277, Ecsery Z, Kosa I, Knoll J, Somfai E, "Verfahren zur Herstellung von neuen,optisch aktiven Phenylisopylamin-Derivaten [Process for the preparation of new, optically active phenylisopylamine derivatives]", published 1970-04-30, assigned to Chinoin Gyógyszer-és Vegyészeti Termékek Gyára RT
- ^ J. Hermann Nee Voeroes, Z. Ecsery, G. Sabo, L. Arvai, L. Nagi, O. Orban, E. Sanfai, U.S. patent 4,564,706 (1986)
- ^ EP 344675, Hájicek J, Hrbata J, Pihera P, Brunová B, Ferenc M, Krepelka J, Kvapil L, Pospisil J, "Method for the production of selegiline hydrochloride", published 989-12-06, assigned to SPOFA Spojené Podniky Pro Zdravotnickou Vyrobu
- ^ Fowler JS (July 1977). "2-Methyl-3-butyn-2-ol as an acetylene precursor in the Mannich reaction. A new synthesis of suicide inactivators of monoamine oxidase". The Journal of Organic Chemistry. 42 (15): 2637–2639. doi:10.1021/jo00435a026. PMID 874623.
- ^ "Sanofi Extends Holding in Chinoin". The Pharma Letter. September 19, 1993.
- ^ a b c d Magyar K (2011). "The pharmacology of selegiline". In Youdim M, Riederer P (eds.). Monoamine Oxidases and Their Inhibitors. International Review of Neurobiology. Vol. 100. Academic Press. ISBN 978-0-12-386468-0.
- ^ Knoll J, Ecseri Z, Kelemen K, Nievel J, Knoll B (May 1965). "Phenylisopropylmethylpropinylamine (E-250), a new spectrum psychic energizer". Archives Internationales de Pharmacodynamie et de Therapie. 155 (1): 154–164. PMID 4378644.
- ^ Magyar K, Vizi ES, Ecseri Z, Knoll J (1967). "Comparative pharmacological analysis of the optical isomers of phenyl-isopropyl-methyl-propinylamine (E-250)". Acta Physiologica Academiae Scientiarum Hungaricae. 32 (4): 377–387. PMID 5595908.
- ^ a b Healy D (2000). "The psychopharmacology of life and death. Interview with Joseph Knoll.". The Psychopharmacologists, Vol. III: Interviews. London: Arnold. pp. 81–110. ISBN 978-0-340-76110-6.
- ^ Birkmayer W, Riederer P, Youdim MB, Linauer W (1975). "The potentiation of the anti akinetic effect after L-dopa treatment by an inhibitor of MAO-B, Deprenil". Journal of Neural Transmission. 36 (3–4): 303–326. doi:10.1007/BF01253131. PMID 1172524. S2CID 38179089. Archived from the original on February 12, 2013.
- ^ a b Cromie WJ (November 7, 2002). "Bodkin is Patching up Depression". Harvard University Gazette. Retrieved September 8, 2007.
- ^ Frampton JE, Plosker GL (2007). "Selegiline transdermal system: in the treatment of major depressive disorder". Drugs. 67 (2): 257–65, discussion 266–7. doi:10.2165/00003495-200767020-00006. PMID 17284087. S2CID 42425086.
- ^ Duffy M (December 3, 2002). "Patch Raises New Hope For Beating Depression". The New York Times. ISSN 0362-4331.
- ^ Cascade EF, Kalali AH, Preskorn SH (June 2007). "Emsam: the first year". Psychiatry. 4 (6): 19–21. PMC 2921248. PMID 20711332.
- ^ Ferdinandy P, Yoneda F, Muraoka S, Fürst S, Gyires K, Miklya I (February 2020). "Geroprotection in the future. In memoriam of Joseph Knoll: The selegiline story continues". European Journal of Pharmacology. 868: 172793. doi:10.1016/j.ejphar.2019.172793. PMID 31743738. S2CID 208185366.
- ^ Knoll J, Miklya I (December 2016). "Longevity study with low doses of selegiline/(-)-deprenyl and (2R)-1-(1-benzofuran-2-yl)-N-propylpentane-2-amine (BPAP)". Life Sciences. 167: 32–38. doi:10.1016/j.lfs.2016.10.023. PMID 27777099.
- ^ "In memoriam Joseph Knoll (1925-2018) | Hungarian Society for Experimental and Clinical Pharmacology". Retrieved April 10, 2023.
- ^ a b Knoll J (January 31, 2018). How Selegiline ((-)-Deprenyl) Slows Brain Aging. Bentham Books. pp. (back of book). ISBN 978-1608055944.
- ^ "Selegiline: Benefits, Dosing, Where To Buy, And More!". February 1, 2023. Retrieved September 2, 2023.
- ^ Carageorgiou H, Sideris AC, Messari I, Liakou CI, Tsakiris S (August 2008). "The effects of rivastigmine plus selegiline on brain acetylcholinesterase, (Na, K)-, Mg-ATPase activities, antioxidant status, and learning performance of aged rats". Neuropsychiatric Disease and Treatment. 4 (4): 687–699. doi:10.2147/ndt.s3272. PMC 2536534. PMID 19043511.
- ^ Stoll S, Hafner U, Pohl O, Müller WE (1994). "Age-related memory decline and longevity under treatment with selegiline". Life Sciences. 55 (25–26): 2155–2163. doi:10.1016/0024-3205(94)00396-3. PMID 7997074.
- ^ Puurunen K, Jolkkonen J, Sirviö J, Haapalinna A, Sivenius J (February 2001). "Selegiline combined with enriched-environment housing attenuates spatial learning deficits following focal cerebral ischemia in rats". Experimental Neurology. 167 (2): 348–355. doi:10.1006/exnr.2000.7563. PMID 11161623. S2CID 22769187.
- ^ Knoll J (May 1992). "The pharmacological profile of (-)deprenyl (selegiline) and its relevance for humans: a personal view". Pharmacology & Toxicology. 70 (5 Pt 1): 317–321. doi:10.1111/j.1600-0773.1992.tb00480.x. PMID 1608919.
- ^ Saunders N, Heron L (1993). E for Ecstasy. London: N. Saunders. ISBN 978-0-9501628-8-1. OCLC 29388575.[page needed]
- ^ Saunders N. "Test results of 30 samples of Ecstasy bought in British clubs between 11/94 and 7/95".
- ^ Pearce D (1995). The Hedonistic Imperative. OCLC 44325836.
- ^ "Sam Barker and David Pearce on Art, Paradise Engineering, and Existential Hope (With Guest Mix) | The FLI Podcast". Future of Life Institute (audio, transcript). June 24, 2020.
- ^ Hurwitz G (2019). Out of the dark. Penguin Books. p. 431. ISBN 9780718185480.
- ^ a b Braddock JA, Church DB, Robertson ID (2004). "Selegiline Treatment of Canine Pituitary-Dependent Hyperadrenocorticism" (PDF). Australian Veterinary Journal. Archived from the original (PDF) on November 29, 2010. Retrieved April 8, 2011. (PDF)
- ^ a b Eghianruwa K (2014). Essential Drug Data for Rational Therapy in Veterinary Practice. AuthorHouse. pp. 127–128. ISBN 978-1-4918-0010-2.
- ^ a b "Anipryl Tablets for Animal Use". Drugs.com. Retrieved August 31, 2017.
- ^ Lundgren B. "Canine Cognitive Dysfunction". Veterinary Partner. Retrieved April 8, 2011.
- ^ a b Riviere JE, Papich MG (2013). Veterinary Pharmacology and Therapeutics. John Wiley & Sons. p. 530. ISBN 978-1-118-68590-7.
- ^ a b c Papich MG (2015). Saunders Handbook of Veterinary Drugs: Small and Large Animal. Elsevier Health Sciences. p. 722. ISBN 978-0-323-24485-5.
- ^ Padilha SC, Virtuoso S, Tonin FS, Borba HH, Pontarolo R (October 2018). "Efficacy and safety of drugs for attention deficit hyperactivity disorder in children and adolescents: a network meta-analysis". European Child & Adolescent Psychiatry. 27 (10): 1335–1345. doi:10.1007/s00787-018-1125-0. PMID 29460165. S2CID 3402756.
- ^ a b Buoli M, Serati M, Cahn W (2016). "Alternative pharmacological strategies for adult ADHD treatment: a systematic review". Expert Review of Neurotherapeutics. 16 (2): 131–144. doi:10.1586/14737175.2016.1135735. PMID 26693882. S2CID 33004517.
- ^ Rubinstein S, Malone MA, Roberts W, Logan WJ (August 2006). "Placebo-controlled study examining effects of selegiline in children with attention-deficit/hyperactivity disorder". Journal of Child and Adolescent Psychopharmacology. 16 (4): 404–415. doi:10.1089/cap.2006.16.404. PMID 16958566.
- ^ Akhondzadeh S, Tavakolian R, Davari-Ashtiani R, Arabgol F, Amini H (August 2003). "Selegiline in the treatment of attention deficit hyperactivity disorder in children: a double blind and randomized trial". Progress in Neuro-Psychopharmacology & Biological Psychiatry. 27 (5): 841–845. doi:10.1016/S0278-5846(03)00117-9. PMID 12921918. S2CID 23234928.
- ^ Wilens TE, Spencer TJ, Biederman J (March 2002). "A review of the pharmacotherapy of adults with attention-deficit/hyperactivity disorder". Journal of Attention Disorders. 5 (4): 189–202. doi:10.1177/108705470100500401. PMID 11967475. S2CID 37417459.
- ^ Tcheremissine OV, Salazar JO (June 2008). "Pharmacotherapy of adult attention deficit/hyperactivity disorder: review of evidence-based practices and future directions". Expert Opinion on Pharmacotherapy. 9 (8): 1299–1310. doi:10.1517/14656566.9.8.1299. PMID 18473705. S2CID 73193888.
- ^ a b c Hailwood, Jonathan Michael; Apollo-University Of Cambridge Repository; Apollo-University Of Cambridge Repository; Bussey, Timothy John (September 27, 2018). Novel approaches towards pharmacological enhancement of motivation (Thesis). pp. 13–14. doi:10.17863/CAM.40216.
- ^ Yohn SE, Reynolds S, Tripodi G, Correa M, Salamone JD (April 2018). "The monoamine-oxidase B inhibitor deprenyl increases selection of high-effort activity in rats tested on a progressive ratio/chow feeding choice procedure: Implications for treating motivational dysfunctions". Behav Brain Res. 342: 27–34. doi:10.1016/j.bbr.2017.12.039. PMID 29292157.
- ^ Contreras-Mora H, Rowland MA, Yohn SE, Correa M, Salamone JD (March 2018). "Partial reversal of the effort-related motivational effects of tetrabenazine with the MAO-B inhibitor deprenyl (selegiline): Implications for treating motivational dysfunctions". Pharmacol Biochem Behav. 166: 13–20. doi:10.1016/j.pbb.2018.01.001. PMID 29309800.